Document Type : Original Research Article
Authors
College of Basic Education, University of Babylon, Babil, Iraq
Abstract
Tissue engineering benefits from electrospun scaffolds, particularly as drug carriers and reconstructive materials for orthopedic implants, as well as many other uses obtaining a large number of publications in a short period in the region through the production of complex scaffolds, the development of new nanotechnology processes, and improvement of imaging methods. Labeling these materials has become critical to achieving accurate and satisfactory results. This is an excellent method for mimicking the extracellular matrix of bone using biodegradable and biocompatible polymers for bone restoration. In this project, electrospinning of a PMMA: PVA scaffold is used. These composite fibers had a clear and continuous shape when examined under a scanning electron microscope (SEM), and their components were identified using (FTIR). Experiments revealed that this characterization of significant effects in the electrospinning method for biomedical applications plays an important role in producing implant coating materials for bone reconstruction.
Graphical Abstract
Keywords
Introduction
Nanofibers should have a diameter of 1 to 100 nanometers. Many factors influence size ranges, including polymer type, solution data, applied voltage, and electrode spinning circumstances. Surface area, porosity, tensile strength, and flexibility are all characteristics of fibers. Because of these features, the structure is a highly robust material employed in a variety of applications including medication release, wound treatment, protective garments, tissue engineering, and sensors. Nanofibers are employed in various sectors, including conductive films to produce chips [1]. The biomedical field is the most important of these applications or radio frequencies, and in electrical device shielding [2]. The fibers' ability is improved by their dielectric characteristic. There is other evidence in the literature [3]. Carbon nanofibers, in particular, have lately been employed in the manufacture of fuel cells [4]. The origins of electrospinning may be dated back to the early 1930s. Morton and Cooley invented the electrospinning technique in 1902, but Formals revived and refined the technology and patented it in 1934. The collector was comparable to a spinning drum in the old technique, and the devices for the spinning process were mobile at the time. Although the method's simplicity has become more important to the world's sciences, it has certain limitations. Among these drawbacks are the inability to dry fibers quickly and insufficient distance between collector and spinning region. The challenges were addressed in the 1940s, when the specified distance was improved and adequate time was allowed to dry and correctly manufacture fibers in various polymer formulas. Taylor researched electrospinning, shaping, and processing during the 1960s. It is worth noting that the polymer takes on a droplet shape when exposed to an electric field, and that this conical shape seen in Figure 1 has been dubbed "Taylor Con" by other researchers [5]. Electrospinning technology is the technique of creating nanofibers by extruding an electrically charged polymer, which causes the fibers to combine into non-woven fabrics [6].
Three basic components of electrospinning are depicted in Figure 2: a DC and pump, and cylinder, or metal plate collector. Between the collector and the syringe, the high voltage electricity produces a lot of electrical energy. When the applied voltage falls below a certain level, the fluid gets charged, forming a Taylor cone. The droplet transforms into a polymer when the surface charge surpasses the liquid's tension. Static electricity causes the injected polymer to dry in flight, resulting in nanofibers on the collector [7].
Nanofiber creation is influenced by a number of factors, including (a) process conditions such as voltage, distance, and feed rate, (b) liquid characteristics such as conductivity, viscosity, and surface tension, and (c) ambient variables such as temperature and humidity [8]. The molecular weight, viscosity, and conductivity of a liquid all influence a polymer's capacity to spin [9]. Nanofibers have lately been explored and are gaining popularity due to their electrical, optical, and electrochemical capabilities, and they are made via electrospinning [10]. Electrospinning technique can readily manufacture varied qualities using polymers. This procedure is simple and cost-effective. Nanofibers may be made from both synthetic and natural polymers, and they can readily imitate the structural features of extracellular matrix (ECM) for bioengineering purposes. New features of nano-sized materials offer benefits and biotechnological applications in nanofiber engineering. One of the most useful nanomaterials is nanofibers. Nanofibers' nonporous nature enables for extensive application of fibers in biomedicine. Natural materials, synthetic polymers, composites, metals, and ceramics can all be used to create porous scaffolds. ECM.15 Cells are grown in vitro on the surface of nanofibers in a tissue engineering field. Engineering scaffolding will support cell adhesion, proliferation, and differentiation. Scaffolding may be made in various ways, although electrospinning and scaffolding based on nanofibers are the most effective [11]. Fibers are used in filtration, optical, agricultural, electrical, biomedical, membrane technologies, and aerospace [12]. In both academics and industry, the dielectric method has become more important and to find the best conditions for producing fibers with consistent morphology and the appropriate functionality from various polymers [13]. PVA is a non-toxic, synthetic, and water-soluble polymer. Many nanofibers are combined with any chemical using electrospinning technology since PVA has strong fiber forming characteristics. PMMA is an FDA-approved food-grade biopolymer. Drug delivery systems, medical procedures, and implants employ this waterproof polymer [17]. Acrylic, or polymathic methacrylate, is a strong, transparent thermoplastic polymer with low density and excellent handling properties utilized in a variety of applications [18]. The novel ECM expression may be done using electrospun nanofibers that completely support cell movement, adhesion, and differentiation. Electrospinning was used to create nanostructures for this research. Scanning electron microscopy (SEM) and Fourier transform infrared spectroscopy are two major approaches that have been used to complete this research (FT-IR). As a result, the qualities necessary for these types of nanostructures for biomedical applications are reviewed in this work, and the nanofiber mat was recapitulated in Table 1 by electrospinning the compound.
Experimental
Material
Polyvinyl alcohol (MW = 84,000 Da) with poly(methyl methacrylate) (MW = 1,10,000 Da) were bought from local laboratory suppliers (Missouri). All chemicals were obtained from the College of Basic Education/University of Babylon laboratories, including ice acetic acid and formic acid without any alteration or purification, as received.
Electrospinning method
Electrospinning was used to make the scaffolds one at a time.
Brief (2:8,4:6,8:2,5:5,6:4) (w/v) poly(methyl methacrylate)/poly(vinyl alcohol) sits on hot plate for 4:5 hours to make a pure nanosolution with high viscosity. The homogenous solutions were then deposited in 5 mL of plastic and secured with a 20 percent needle. A high-voltage power supply of 10 kV was used. The needle is eight centimeters away from the target. Using a rotary syringe pump with a collector speed of 400 rpm, the flow rate of the polymer solution is changed at a rate of 0.5 ml/hr. On a cylinder-shaped nano-scaffolding complex, the nanofibers were produced by spinning titanium. The micro-optic process is schematically depicted in Figure 3.
Characterization
After SEM identification of the scaffolds, the nanofiber shape was determined (FEI Quanta FEG 250). A small layer of gold was sprayed on the nanofibers that suited the device and were coated with conductive film. Poly (methyl methacrylate)/poly (vinyl alcohol) was investigated using FTIR (Bruker Vertex 80-HeNe Laser) across the wavenumber range 500-4000 cm-1, in addition to the chemistry and analysis of dry electrospun nanofibers. The fiber size was determined using ImageJ program to create nanoparticles.
Results
SEM analysis
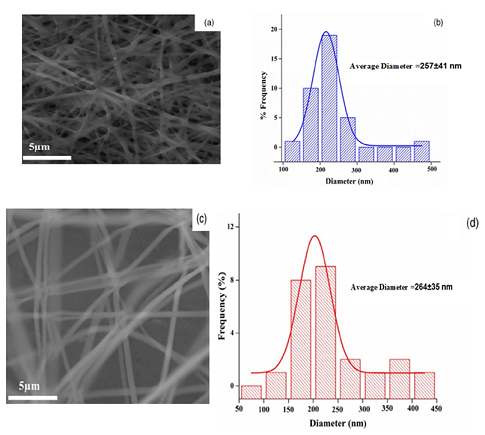
A nanofiber mat containing 25% PMMA and 10% PVA was created to load placebos and medicines. For PVA and PMMA, the SEM pictures are fake nanofibers. Fibers with smooth surface forms were distributed in a semi-uniform fashion. As indicated in Figure 4, the SEM images of a PVA and PMMA nanofiber mat (a) and (c) demonstrate a homogeneous distribution and varied surface shape. If the medicine is integrated into the nanofibers, this change in conformation may occur. Image J software was also used to determine the average diameter. 257± 41 nm (PVA: PMMA) and 264±35 nm were discovered (PMMA:PVA). Therefore, nanofibers were made of PVA and PMMA. The PVA:PMMA (v/v) ratio was varied from 2:8 to 8:2. The lowest diameter was seen in a 5:5 ratio of PVA:PMMA in SEM pictures of these structures, which clearly displayed simple shape and consistent distribution. As a result, the 5:5 ratio was produced, and it may be widely employed to create nanofibers for drug loading. Figure 4 (e) illustrates the SEM images of a composite nanofiber mat (PVA:PMMA) demonstrating the dense network of fibers. In addition, the nanofibers' basic shape may be preserved to the mat, which has an average diameter distribution of 244±81 nm.
FTIR analysis
The existence of an OH group, which is a physiologically active group, is indicated by the emergence of a peak at 3552. Furthermore, the existence of a peak at 2989 indicates the presence of the CH3 group found in the polymers PMMA:PVA. The existence of a peak at 2949 indicates the presence of the CH2 group, which is also seen in the polymers PMMA:PVA and the existence of a C=O group in the polymer PMMA, as evidenced by the emergence of a peak at 1728. The existence of a C–O group in the polymer PMMA:PVA is evidenced by the formation of a peak at 1192, which is characterized by medicinal and biological qualities, as displayed in Figure 5. Blend's research aims to improve the characteristics of polymers in medicinal applications.
Discussion
Nanotechnology is the process of designing systems and manipulating materials on a massive scale using nanometer-scale dimensions. This approach adds definition to the material, which has a wide range of qualities and may be used for various biological applications. The major goals of tissue engineering in transdisciplinary domains are also to repair and manipulation, or replace injured tissue with functioning cells, biomaterials, and blood vessels, and use the physical qualities of biomaterials to facilitate the regeneration process [19]. For these objectives, one of the characterization processes of PVA and PMMA are combined as two electrolytic polymers. Recent studies [20-24] have found that fine nanofibers are useful in bone engineering. It is an excellent tissue producer as well as a "smart" bone remodeling substance. Electrospinning was used to create nanofibrous scaffolds, which are the best to employ in the form of "smart" hair follicles, according to our present research. After studying the nanofiber architectures separately, pure nanofibers made of PMMA:PVA were produced. Fiber morphology was created with more details. SEM morphology is depicted in Figure 4. Nanofibers with little grain formation, as shown in SEM images, have a detrimental impact on porosity. Nanofibers are extremely appealing for biological applications because of this condition. When minute grains the first emerge, here is where they originate. The beads, however, are quite minute and have little influence on the overall morphology of smooth fibers.
Conclusion
Most bacterial infections develop biofilm as a result of biological drug resistance. The key is focused medication delivery, which helps increase therapeutic dosage at the treatment location while minimizing unwanted effects. Electrospinning technique improved and made electrically insulated nanofiber mat (PVA, PMMA, and PVA: PMMA). We deal with various PVA and PMMA (2:8 to 8:2) combinations that have been created before employing the electrospinning method. The smallest proportion in determining its diameter can be combined with other components in a polymer solution for medication delivery and other purposes. When compared to other nanofibers, three distinct forms of composite nanofibers had the best fibers. The nanofiber medication release is dependent on the percentage of tumor that the nanofiber is exposed to the buffer media and the compound turned out to be 280 percent for 24 hours, making it more flexible and pleasant. The medication is released in accordance with the first-order movement. The activity of all antibacterial nanofiber mats was compared. These fibers, which are also physiologically active, are bactericidal for biofilm problem.
Acknowledgements
The authors thank their universities for their support.
Conflict of Interest
The authors declare no conflict of interest.
Orcid:
Qasim S. Kadhim: https://orcid.org/0000-0002-5254-4830
Ali L. Alfalluji: https://orcid.org/0000-0003-1876-2444
Fadhel O. Essa: https://orcid.org/0000-0001-9398-3605
------------------------------------------------------------------
------------------------------------------------------------------
Copyright © 2023 by SPC (Sami Publishing Company) + is an open access article distributed under the Creative Commons Attribution License(CC BY) license (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

.png)
.png)
.png)
.png)
.png)
.png)
.png)
