Document Type : Original Research Article
Authors
1 Physics and Chemistry Group, Faculty of Basic Sciences, Imam Ali University, Tehran, Iran
2 Department of Chemistry, Faculty of Science, University of Zanjan, Zanjan, Iran
3 Department of chemistry, Faculty of sciences, Imam Ali University, Tehran, Iran
4 Department of Chemistry, University of Zanjan, P O Box 45195-313, Zanjan, Iran
5 Department of Biotechnology, Research Institute of Modern Biological Techniques (RIMBT), University of Zanjan, Zanjan 45371-38791, Iran
Abstract
The new type of coronavirus called Severe Acute Respiratory Corona Virus-2 (SARS-CoV-2) has spread over the globe, left the world in lockdown, and taken more lives globally. Hence, more countries have taken restricted policies including quarantine to protect people. However, this was not a promising agent to tackle the existing crisis, so it could become burden on the scientists’ shoulders to come up with noble ideas and bring high-safe vaccines and antiviral drugs to combat the COVID-19 pandemic. So far, vaccines and drugs like paxlovid and molnupiravir have been developed to effectively change the pandemic course if clinical trials will prove their efficiency. However, no FDA-approved antiviral drugs against SARS-CoV-2 yet exist. To gain these types of antiviral drugs, computer-aided drug design techniques play a notable role. Of these approaches, molecular docking can screen bio-active compounds and test their potential to specific targets. Herein, we screened bio-active compounds of royal jelly and honey against SARS-CoV-2 main protease in order to investigate their antiviral potency conducting in vivo and in vitro tests. Among the tested natural products, 4 compounds, namely Erlose, Kaempferol glucoside, Iridin, and luteolin glucoside (Cynaroside) showed the lowest binding energies of -10.2, -9.6, -9.0, and -8.4 kcal/mol, respectively. Then, two marketed drugs, i.e. Indinavir and lopinavir, were chosen as standard drugs. Among all, Erlose and Kaempferol glucoside represented excellent antiviral peculiarities against COVID-19 main protease, and may be on the call for future clinical trials to approve their potency.
Graphical Abstract
Keywords
Introduction
In recent decades, viral diseases including H1N1 influenza, SARS-COV-1, COVID-19 and Ebola have threatened the health of human society. In late 2019, a new virus from the corona virus family called SARS-CoV-2 appeared in Wuhan, China. Since then, Covid-19 disease has spread rapidly around the world and has taken millions lives around the globe [1]. SARS-CoV-2 virus belongs to the beta-coronavirus family viruses, bearing about 93% genetic similarity to the virus present in the bat [2]. This virus contains 4 main proteins including E, M, S, and N. SARS-CoV-2 Spike glycoprotein attaches to the host cell receptor named Angiotensin-Converting Enzyme 2 (ACE-2) and simplifies the cell fusion process in which viral RNA goes through the cell and viral infection can be facilitated [3]. Discovery of proficient antiviral drugs for SARS-CoV-2 vaccines is currently the most significant challenge for research groups. Though a couple of vaccines hit the market and around half of inhabitants in developed and developing countries got vaccinated, the death rate would not become zero, meaning that finding an effective antiviral drug for the current viral disease needs a great deal of attention and hardworking [4,5]. The use of natural compounds with antiviral properties is of great importance due to the ease of production on a large scale and the wide range of available compounds [6-8]. Performing simulations through computational chemistry software [3] with the aim of finding drugs with antiviral properties is considered as the first step in evaluating the effectiveness of these natural components [3]. The biological properties of a large number of compounds can be predicted using computational techniques and can be screened for experimental experiments [9,10]. Using natural compounds possesses some benefits, including being non-toxic, green, environmentally-friendly, and having supreme biological activities [11,12]. One of the most important natural ingredients is royal jelly, a honey bee product and a creamy, thick, milky white substance with a special taste and smell that results from the secretions of the pharyngeal glands in the head of young nurse worker bees [13] with high nutrition importance fed to adult queens and larvae. Incomplete digestion of honeydew in the stomach of worker bees can produce this secretion [14]. Royal jelly is of high significance nutrition for larvae and can cause a discrimination in the caste of these species: Larvae that receive royal jelly for about 3 days will become queens although those larvae designated to become worker bees will consume pollen, water, and honey in a mixture. Finally, worker bees survive up to a couple of months while honeybee queen lives for a longer time (up to several years). This longer period of queens’ lives refers to the excellent nutrition of royal jelly [15]. Royal jelly is composed of minerals and vitamins in low amounts, proteins (12%), sugar (12%-15%), and water (60%-70%) [14].
Royal jelly can strengthen the immune system [16] and bears powerful peculiarities such as antimicrobial [16], hormone regulatory, blood pressure regulatory [17], blood lipid depressor [16], metabolism regulatory [18] and etc. Isolated peptide from royal jelly also depicted suitable antibacterial and antifungal peculiarities [19]. This systematic review noted that these peptides may protect those individuals infected with SARS-CoV-2 from co-infectious [19]. Royal jelly also postulated possible antiviral activity against HSV-1[20] and in vivo studies on these proficient natural product revealed prophylactic factor owing to its benefits to immune system. Their results highlighted that the mice received royal jelly supplementation showed a larger production of immunocompetent cell proliferation and antibodies, pointing out that royal jelly and its compound with supreme prolypheratic and therapeutic may be a potential candidate against SARS-CoV-2 [21].
Concerning antimicrobial activities of royal jelly, recent works attenuates that it may have a strong effect on growth of fluconazole-resistant fungal strains and also antibacterial against Gram-positive bacteria and low effect against gram-negative bacterial strains [22,23]. Royal jelly containing phenolic or non-phenolic compounds such as kaempferol, Luteolin, Iridin, and erlose with considerable biological activities can be briefly explained as follow. Kaempferol is a flavanol; past studies have affirmed obvious natural highlights of kaempferol and its subordinates e.g., conceivable part within the treatment of viral diseases particularly SARS-CoV-2 [24] as well as blocking power against 3a channel of SARS-CoV [25]. Moreover, preeminent antiviral potential against distinctive viral illnesses named pseudorabies infection [26], HSV-1 [27], Dengue Infection and Japanese Encephalitis Infection (JEV) [28]. These logical records show their strong bioactivities and may ensure the presumptions to in silico and in vitro test against SARS-CoV-2.
Erlose is a trisaccharide consisting of sucrose and can be found not only in honey but also in royal jelly (Figure 1), bearing significant antibacterial properties [29]. Luteolin and iridin are the other high potent bioactive compounds in royal jelly, bearing health benefits, for instance, Luteolin and its analogous have drawn much attention due to their high antiviral potential against Epstein-Barr virus (EBV), HIV-1 [30], and JEV [31]. Iridin also is an existing flavonoid in royal jelly, indicating good antioxidant, antitumor, and anti-proliferative activities. It also showed great antitumor potency to combat AGS gastric cancer cells growth [32].
Honey is the other significant nourishment of honeybee natural products, which can be prepared by a cascade of actions by honeybees for example after collection of floral nectar and plants’ deposits. Mixing with especial substances of honeybees, it will be processed and stored for repining [33]. Honey comprising carbohydrates, acids, and flavonoids in general, posing promising biological features like anti-inflammatory, pro-apoptotic, immune-modulatory, anti-metastatic, and anti-proliferative peculiarities [34]. Honey also shows excellent bioactivities such as wound healing, antioxidant, and antibacterial features [35]. Honey along with propolis may protect against different viral diseases like human T-cell leukemia lymphoma virus type 1 (HLTV-1) [36], influenza virus [37, 38], respiratory syncytial virus (RSV) [39], and Newcastle disease virus (NDV) [40].
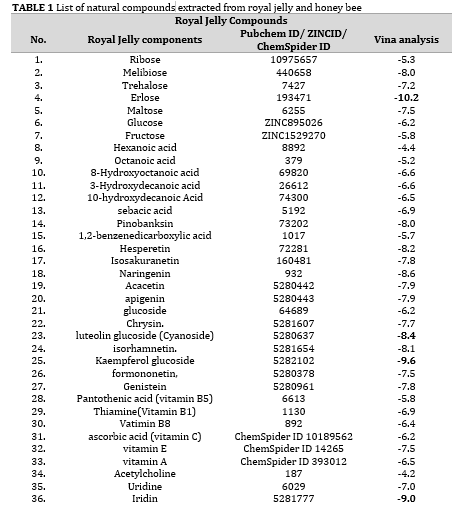
As royal jelly and honey bee naturally occurring compounds have excellent bioactivities, they can be the best choice against SARS-CoV-2 main protease. In this research, we have investigated the antiviral potential of these natural compounds against SARS-CoV-2 main protease using Auto Dock Vina to explore novel natural inhibitors (Table 1). We have chosen Ivermectin and Lopinavir as standard drugs in order to compare their antiviral potency against SARS-CoV-2 main protease with those of natural compounds.
Experimental
Preparation of ligands and receptor
The 3D structures of natural compounds were downloaded from PubChem and Zinc 15, and ChemSpider databases in SDF format and converted into PDB using open babel [1]. The 3D crystal structure of SARS-CoV-2 main protease (PDB code: 6LU7) was also extracted from Protein Data Bank(PDB) [2]. Autodock Tools (ADT) (1.5.6) was utilized for pre-processing steps [3]], e.g., adding the polar hydrogens, atom types of all protein and ligands, and gastiger charges. In terms of ligands preparation, the torsions were set to be active to develop searching phase space. Then, the ligands were saved in PDB for AutoDock Vina.
Molecular docking via Autodock vina
Molecular docking was fulfilled using Vina 1.1.2 on Windows 10 platform (64-bit) with Lenovo IdeaPad L340-15IWL (CORE i7, system memory: 256 GB SSD, 12 GB RAM). The grid box dimension was set on the active site of SARS-CoV-2 Main Protease with a grid spacing of 1.0 angstrom [4]. The other parameters were kept as default. According to Autodock Vina results, the compounds with the lowest binding energies were ranked as the best possible inhibitors against COVID-19 main protease. Finally, the visualizations ligands with the target were obtained by Discovery studio v 4.5.
Results and discussion
Docking validation
Docking process was validated through re-docking of N3 inhibitor onto binding site of COVID-19 Main Protease (PDB code: 6LU7). It showed a suitable binding energy of -13.4 kcal/mol with making 13 H-bonds with the receptor (Figure 2).
Molecular docking results
Molecular docking is a powerful technique by which affinity of ligands to different protein targets can be investigated [3]. Herein, we have employed AutoDock Vina for investigation of the antiviral potential of natural products extracted from honey bee and royal jelly against SARS-CoV-2 main protease. Moreover, we have selected two marketed drugs (Indinavir and Lopinavir) as standard drugs to compare their results with those of natural inibitors. Top 4 natural inhibtors with the lowest binding energies along with their properties and interactions are listed in Tables 2 and 3.
Among these natural inhibitors, Erlose (compound a), as a remarkable trisaccharide, exists in royal jelly bearing suitable antibacterial properties [5]. It showed the lowest energy among all tested compounds (-10.2 kcal/mol), and also possessed the highest number of H-bonds (7) with LEU 141, GLY 143, SER144, CYS 145, GLU 166, GLN 189. It is noteworthy pointing out that this natural compound could block CYS 145, as one of the main amino acid residue of SARS-CoV-2 main protease, through H-bonding. Although it bounded to the viral strain via one unfavorable donor-donor bond, which can weaken its antiviral potential per se, showing the lowest binding energy and the highest H-bonding with the protease may approve the highest inhibitor potency of this natural compound against COVID-19 (Figure 4a).
As for kaempferol glucoside (compound b), as a flavanol indicating potent antiviral potential against Pseudorabies infection, HSV-1, Dengue Infection and JEI [6-9], and Iridin (compound c), found in royal jelly and delinates significant antirumor potential to fight AGS gastric cancer cells growth, along with suitable anti-proloferative, and antioxidant properties, ranked second and third in terms of binding modes with binding energies of -9.6 and -9.0 kcal/mol, respectively.
Compound c revealed lower binding energy compared to that of compound b and possessed higher number of hydrohen bonds (6) with THR 26, GLY 143, SER 144, HIS 163, GLU 166, GLN 189; compound b showed higher antiviral capabilities, dedicates higher binding energies, the last but not least, blocking two considerable residues of SARS-CoV-2 Main protease , CYS 145 and His 41 (Figure 3) through hydrogen bond and π-π T-shaped interactions. These data may back up the superiority of inhibitory effect of compound b compared with those of the other natural compounds and even compound a (Figure 4b, c).
Another considerable existing compound in royal jelly is luteolin glucoside (compound d), bearing potent antiviral abilities to combat Japanese encephalitis virus (JEV), EBV (Epstein-Barr virus), and HIV-1 [10,11]. This compound showed the lower binding energy with the receptor (-8.4 kcal/mol) compared to those of the other natural inhibitors, indicating 5 H-bonds with THR 24, THR 25, GLY 143, and SER 144 along with a Pi-alkyl interaction CYS 145. Apart from the convincible number of H-bonds with protein target, compound d illustrated two unfavorable bonds, which can significantly diminish its antiviral potential compared to the other tested natural compounds (Figure 4d). Two marketed drugs Indinavir (compound e) and Lopinavir (compound f) were chosen as standard drugs. These two control drugs exhibited binding modes of -7.7 and -7.5 kcal/mol, respectively. As can be seen in Figure 4e, f, there is an energy difference of 1-3 kcal/mol in average (in terms of binding energy) between natural inhibitors and marketed drugs, postulating the higher peculiarities of these natural inhibitors in comparison to their synthetic standard counterparts. Another point that signifies the superiority of natural inhibitors over their synthetic standards is that compounds e and f possessed much lower number of H-bonds, just two for compound e and one H-bond for compound f. Finally, these standard drugs made lower electrostatic interactions compared with natural components (Compounds e and f). It can be concluded that natural inhibitors bore high superiority versus their synthetic counterparts, according to molecular docking data.
Conclusion
As no promising treatment exists for SARS-CoV-2 viral disease to date, more industrial companies globally still are making hard endeavor to discover high potential antiviral drugs to stop the current horrible COVID-19 outbreak, like paxlovid and molnupiravir drugs as examples to have a prominent effect against COVID-19 if clinical trials approve their antiviral impact by near future. This research shed light on antiviral affinity of bioactive natural compounds extracted from royal jelly and honey bee against SARS-CoV-2 MPro (PDB code: 6LU7) using molecular docking study through Autodock Vina. Molecular docking assessments were supplemented using Vina, and 3D Visualizations of natural ligands in complex with the target protein were taken by Discover studio v 4.5. In order to investigate and compare the antiviral potential of these natural products with the affinity of marketed drugs, two standard drugs, i.e. Indinavir and Lopinavir, were selected. 4 natural compounds out of all, namely Erlose, Kaempferol glucoside, Iridin, and luteolin glucoside (Cynaroside) illustrated the lowest binding energies of -10.2, -9.6, -9.0, and -8.4 kcal/mol, respectively. Concerning the available information from molecular docking, compounds a and b can be hopefully nominated as promising antiviral candidates against SARS-CoV-2 infectious if they are successfully passed through in vitro and in vivo studies to prove their antiviral potential.
Acknowledgements
The Authors would like to appreciate Imam Ali University for its support.
Orcid:
Rasool Amirkhani: https://www.orcid.org/0000-000-2330-4598
Armin Zarei: https://www.orcid.org/0000-0003-2326-4431
Mahdi Gholampour: https://www.orcid.org/0000-0002-9553-4894
Hassan Tavakoli: https://www.orcid.org/0000-0002-9037-8300
Ali Ramazani: https://www.orcid.org/0000-0003-3072-7924
---------------------------------------------------------------------------
How to cite this article: Rasool Amirkhani*, Armin Zarei*, Mahdi Gholampour, Hassan Tavakoli, Ali Ramazani. Honey and royal jelly natural products as possible antiviral nominations to combat SARS-CoV-2 main protease. Eurasian Chemical Communications, 2022, 4(6), 567-579. Link: http://www.echemcom.com/article_147345.html
---------------------------------------------------------------------------
Copyright © 2022 by SPC (Sami Publishing Company) + is an open access article distributed under the Creative Commons Attribution License(CC BY) license (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

.png)
_(1).png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
.png)
