Document Type : Original Research Article
Authors
1 Department of Chemical Engineering, Farahan Branch, Islamic Azad University, Farahan, Iran
2 Barsad Sazan Kimiya Company, Tehran, Iran
Abstract
Today, due to the industrialization of human societies and the need to use fossil fuels as much as possible, the face of most cities in the world has become black and dirty due to pollution and fats from these fuels. In proportion to the progress of societies, the discovery of nano-sized materials can be the solution to many problems. Among them, two types of self-cleaning and hydrophobic coatings have been developed, which are based on hydrophobic properties, but with the difference that self-cleaning coatings are possible. Clean surfaces only by sunlight. By creating a coating of this material on the exterior of buildings and various structures, it is possible to decompose existing pollution, which are mostly hydrocarbon compounds, only by using sunlight. In addition to creating anti-water properties, nano self-cleaning solutions are very effective with the use of statin light that removes dust from surfaces. One of its applications is to use as anti-fog and self-cleaning materials. The hydrophilic properties of its materials allow water to spread completely on the surface instead of remaining as small droplets. Therefore, it can achieve anti-fog and self-cleaning effects. In these compounds, the catalyst can have self-cleaning and anti-fog effects. The complementary combination of PEG and 2SiO2 can improve such catalyst efficiency as crystal structure, crystal size, porosity level, and surface area.
Graphical Abstract
Keywords
Introduction
The use of nanotechnology can create important and new applications in various fields of agricultural sciences in such a way as to improve plant performance, increase growth and photosynthesis. Product management practices can be improved by using this technology [1-3]. The rate of absorption of nanoparticles in plants is different and depends on the type of plant, chemical composition, and size of these particles. Titanium dioxide nanoparticles (TiO2) are widely used in agricultural industries to increase the growth characteristics of plants [4-6]. With the advancement of nanotechnology and the wide applications of titanium dioxide nanoparticles in various industries, there is concern about their toxic effects on the environment and human health [7-9].
By creating new materials, nano technologies have provided new hope for environmental engineers to deal with environmental pollution [10]. However, the exposure of consumers and sensitive groups to them increases the concern about their production and use. By passing from microparticles to nanoparticles, some physical properties will change, including the surface-to-volume ratio and reaching the domain and quantum effects. The increase in surface area relative to the volume causes the behavior of external atoms to be superior to internal atoms.
Nanoscale catalysts increase the efficiency of chemical reactions and combustion, and at the same time greatly reduce pollution and waste materials. Today, more than half of the useful drugs on the micron scale are not soluble in water, and if they are, most likely, if they are in nanometer sizes will be dissolved in water. These points are only a limited number of the advantages of making materials at the nano scale [11].
In this article, an experimental investigation of the effect of titanium nanoparticles on the properties of hydrophobic self-cleaning film was investigared and basically at the end, when we talk about investigating the properties of a nanocatalyst, it means that either the activity of the catalyst, or the selectivity of the nanocatalyst has been investigated, or research has been done to understand the mechanism of the nanocatalyst's performance.
Research literature
TiO2 is chemically very stable, inexpensive, and the holes created in it by light irradiation are highly oxidizing. The holes created by light irradiation, after reacting with water, these holes generate •OH radicals and its potential decreases to a small amount. The reduction potential for the conduction band of electrons is -0.52. Therefore, it is negative enough to reduce oxygen molecules as O-2. According to the existing conditions, holes O-2, H2O2, O2, •OH radicals can play an important role in the mechanisms related to the photocatalytic process. In relation 1, the reactions related to the photocatalytic mechanism of TiO2 are given [11].
Physical properties of nano titanium dioxide
Crystal structure
Titanium dioxide exists in three main phases (three crystalline phases): rutile, anatase, and brookite. Among these three phases, the rutile phase is more stable and the other two phases, anatase and brookite, are converted into rutile due to heat. Spatially, rutile and anatase phases are tetragonal and brookite is orthorhombic. For some applications, such as normal solution filtering, the crystal phase is not needed. The crystalline phase is necessary when a specific application such as photocatalytic or semiconductor is considered. For example, anatase phase is used for photosensitive dyes and photocatalysis, and rutile phase is mostly used in dielectrics and high temperature oxygen sensors [12].
TiO2 Hydrophilicity
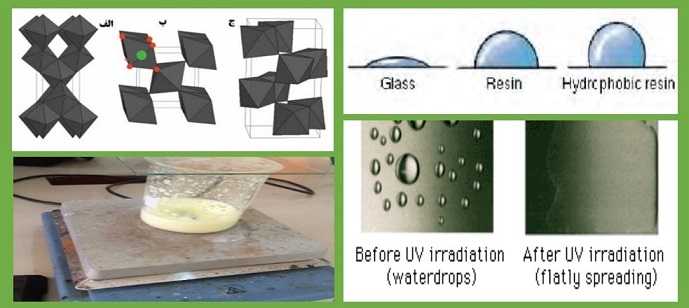
The superhydrophilicity properties of the surface allow the water to spread completely on the surface so that it remains as a drop. As a result, glasses with Tio2 coating become anti-fog and self-cleaning [13-15]. The ability to control the wettability of material surfaces is important in many places. The characteristic of these surfaces is attributed to the combination of the microstructure of hydrophilic and oil-loving phases that are obtained under UV radiation. After irradiation, water droplets spread on the film and produce a contact angle of about 0 degrees. Such a change in wettability is more obvious when TiO2- coated glasses are exposed to water vapor. Hence, before UV light irradiation, water droplets are created on the glass and after the radiation, no trace of them was observed and the glass is transparent [16].
Superhydrophilicity
In our living environment, the surface of a material repels water droplets up to several degrees. The degree of water absorption from a material is defined as the contact angle of the water droplet with the surface. On glass or other mineral materials, a drop of water has a contact angle in the range of 20 to 30 degrees. On the surface of plastics, this contact angle is usually between 90 and 70 degrees. Water contact can be more than 90 degrees. A thin layer composed of titanium dioxide, which is combined with appropriate additives, initially shows a contact angle of several tens of degrees. When this surface is exposed to the UV light, the contact angle of the water on it starts to decrease and the water tends to spread on the flat surface instead of becoming a droplet. Finally, the contact angle of the water drop reaches zero degrees. In this case, the surface is completely non-hydrophobic, which is called super-hydrophilic.
Hydrophilic mechanism of TiO2
Titanium dioxide is a substance that can intensify its hydrophilicity under certain conditions. If the surface of this material is stimulated with ultraviolet light, the oxygen bonds of this material will be broken in the vicinity of water and will turn into hydroxyl bonds. Therefore, the titanium atom on the surface has two hydroxyl groups and can absorb water molecules with hydrogen bonds. Therefore, the surface of this material becomes superhydrophilic. In this case, the titanium dioxide surface establishes a weak Van der Waals bond with organic molecules, which are generally non-polar, and the adhesion of these materials to the surface is low. Therefore, the surfaces covered with this material become less dirty due to the adhesion of organic molecules, or after being dirty, contact with water can lift these molecules from the surface and clean it again [17-19].
Relationship between photocatalysis and hydrophilicity of TiO2
The mechanism of these two processes is different. Although in some parts of the reaction mechanism, these two properties are similar to each other, but the photocatalytic properties and hydrophilicity of TiO2 are different from each other. In fact, after the production of electron and hole (similar part of the two properties) due to light radiation, two different reactions may occur. The reaction in which the generated electron-holes react with water and air oxygen and produce hydroxyl radical and superoxide anion radical, leads to the photocatalytic property of TiO2 and the reaction in which the generated electron-hole reacts with TIO4+ cations. And finally, they produce oxygen vacancies, which leads to the hydrophilicity of TiO2. These two reactions do not conflict with each other and both can happen on the same surface at the same time. That is, on one surface, both photocatalytic properties and hydrophilic properties can exist at the same time. Depending on whether the photocatalytic reaction is carried out more (has more efficiency) or the reaction related to hydrophilicity, a surface can have more photocatalytic properties and less hydrophilicity or vice versa. The mutual effect of photocatalysis and hydrophilicity can be explained in this way that due to more hydroxyl groups that can be absorbed on the surface due to hydrophilicity, photocatalytic activity increases, and therefore hydrophilicity can increase photocatalytic activity. On the other hand, the surface of the film has the ability to absorb contaminated compounds, which turns hydrophilic surfaces into hydrophobic surfaces. The photocatalytic feature decomposes the organic compounds on the surface and leads to restoring the hydrophilicity, and in this sense, the photocatalyst improves the hydrophilicity and maintains this feature for a long time. A surface that is only hydrophilic cannot maintain its self-cleaning effect forever. Photocatalyst plays an important role in the survival of self-cleaning in the following two ways [20]:
- Decomposition of compounds contaminated with H2O and CO2 and organic compounds to obtain the self-cleaning effect directly [21].
The effect of PEG and SiO2 on titanium dioxide film
The crystal shape of titanium dioxide film is changed by SiO2 and PEG. PEG can increase the hydrophilic properties and change the crystal size and reduce the possibility of the film cracking. Likewise, SiO2 can increase the acid strength of the catalyst. It can improve the hydrophilic properties of the catalyst even without irradiation. The addition of PEG and SiO2 in the film improves the superhydrophilic properties, which can be used for anti-fogging and self-cleaning in a low-irradiation environment [22-24].
Cover
Hydrophilic and superhydrophilic surfaces
Unlike hydrophobic and superhydrophobic surfaces that only use the mechanism of water droplet flow for the self-cleaning effect, hydrophilic surfaces destroy the chemical structure of pollutants in the vicinity of sunlight. This process is called photocatalysis. This technology is basically inspired by the photosynthesis of green leaves of trees. One of the materials that is widely used as hydrophilic self-cleaning surfaces is titanium dioxide. In 2001, Pilkington glasses were the first self-cleaning coatings to be used commercially; These glasses were made of a thin and transparent layer of titanium dioxide. Titanium dioxide cleans glasses through two distinct mechanisms, photocatalysis and hydrophilicity. During the photocatalysis process, the chemical structure of organic pollutants and the other impurities on the coating is broken by absorbing sunlight. The hydrophilic property of the surface also causes a sheet of water to be created on the surface by reducing the contact angle, which removes pollution. Under normal conditions, titanium dioxide absorbs light that has an energy equal to or greater than its energy gap, hence, this energy creates charge carriers: positively charged holes (+h) and negatively charged electrons (-e).
Titanium oxide is not very active in the visible region and is only able to absorb 4% of sunlight. Titanium oxide usually exhibits superhydrophilic properties under UV light. By adding metals such as chromium, manganese, cobalt, and iron to titanium dioxide nanoparticles, they were able to perform the process of photocatalytic degradation of pigments with the help of these nanoparticles under visible light conditions [24]. When titanium oxide polycrystals are doped with cobalt, due to the intercalation of cobalt ions in the titanium oxide network and the creation of new bonds, the energy gap in titanium oxide decreases; electrons in the network of this crystal can be transferred to the conduction band by absorbing light in the visible region and perform photocatalytic activity in titanium oxide, while the addition of chromium to the structure of titanium oxide can lead to a decrease in the optical absorption edge and an increase in absorption in the visible region [25].
The superhydrophilicity of titanium dioxide is also induced through sunlight. The holes created in titanium dioxide after light stimulation take electrons from the oxygen on the surface of the material and cause its oxidation. Therefore, oxygen free radical is created on the surface, which allows hydrogen bonding. By forming bonds between oxygen and hydrogen radicals, hydroxide groups are formed on the surface, which reduce the contact angle [23-25]. The photocatalytic activity of titanium dioxide increases when we use it as a thin nanocrystalline layer. In addition to titanium oxide, other materials such as WO3, CdS, ZnO, and ZrO2 are also used as self-cleaning surfaces. Since the properties of the surface of the material are different from its interior - because the atoms and molecules that are in the vicinity of the surface of the material have fewer bonds than the atoms inside the material and are more unstable in thermodynamic terms - this instability increases the energy of the material. In addition, the energy levels on the surface are discrete, while inside the material these levels are compressed to form an energy band structure.
Application of superhydrophobic surfaces
One of the most important applications of hydrophobic surfaces is their use as antifreeze. In cold regions, ice is deposited on the surface of solids and causes a drop in the mechanical properties of solid systems. Superhydrophobic surfaces prevent the formation of ice on solids [26-28]. Research results show that the air in cold surfaces should be supersaturated to start the germination of ice crystals. The degree of supersaturation depends on the amount of surface energy, which affects the initial germination of the ice crystal. Cold surfaces that have less energy have a higher degree of supersaturation for germination. Thin hydrophobic layers as surfaces with the lowest surface energy can prevent the formation of ice on solids. The adhesive and magnetic properties of water droplets [29], the interaction of a water droplet and a solid surface, and the repulsion of oil droplets are among the research fields in superhydrophobic surface technology [29].
Self-cleaning and super hydrophobic
This property is achieved by chemical and geometric modification of the textile surface. Creating nano and micrometric irregularities by using a water-repellent coating causes the contamination of the textile surface to easily slip and separate from the water-repellent fabric in the presence of water. In this way, the textile surface remains clean. The idea of creating such a product was inspired by the natural structure of the lotus leaf [30].
Challenges of developing superhydrophobic surfaces
Despite the extensive efforts and research in the field of producing super-hydrophobic textiles with self-cleaning properties, there are challenges in this field that are often related to wear and tear and instability of properties. The nanoscale fluffy structure of textiles is fragile and easily damaged by pressure or even mild wear. Therefore, it seems necessary to create structures that are more stable against incoming forces. Another problem of these surfaces is the tendency to absorb pollution, including oily substances. Pollutants penetrate into the textile structure and are difficult to remove. The mentioned disadvantages make it difficult to use this type of textiles in the long term [31].
Comparison of superhydrophobicity and Tio2 in self-cleaning
The effect of photocatalytic self-cleaning with Tio2 has several advantages over self-cleaning with the superhydrophobic method.
- Easier to use Tio2 than superhydrophobic surfaces.
- Longer durability and strength of surfaces treated with this Tio2.
- Multipurpose finishing using self-cleaning Tio2, anti-bacterial, anti-odor and UV absorber.
- The stability of washing, abrasion, and more suitable environment of textile completed with Tio2.
Research background
In 2011, Dewi et al. worked on changing the crystal shape of Tio2 nanoparticles with PEG and SiO2 for self-cleaning and anti-fogging applications. In this solution, PEG and SiO2 were used simultaneously and added to the titanium dioxide photocatalyst, which the use of precursor TiAcAc is prepared. Adding this additive in the composition changes until reaching the optimal conditions from the characteristics of SEM, XRD, BET, and FTIR, as well as the activity test using contact angle measurement, the optimal condition is with 15% PEG weight and 30% added SiO2 was obtained. In this combination, the catalyst can perform self-cleaning and anti-fog effects even in minimal light. The complementary combination of PEG and SiO2 can improve the efficiency of the catalyst such as crystal structure, crystal size, porosity level and surface area [32].
Shirin Naqvi et al. investigated the photocatalytic and superhydrophilic activity of Tio2 nanoparticle coatings on the surface of glazed tiles for self-cleaning applications and were investigated for the applications of self-healing. The initial solution for the preparation of a stable cell was prepared from certain amounts of titanium alkoxide along with the required additives, then the samples were coated using the immersion technique. The coated tiles were subjected to heat treatment for one hour, and the heat treatment temperature was selected between 500 and 600° using TG and DSC thermal analysis. The crystalline phases formed at different temperatures and topography of the coating surface were evaluated using X-ray diffraction technique and atomic force microscope (AFM). The photocatalytic activity of the coatings was measured by the amount of decolorization of the Nile Blue and the decomposition of stearic acid, which were used as a pollutant, under UV light, respectively, using a UV-Visible device and a Fourier transform infrared (FTIR). The super-hydrophilic property of the coating was also checked by measuring the contact angle of the water droplet before and after UV light irradiation. The results showed that the optimum heat treatment temperature is 600° and the average size of crystals at this temperature is about 15 nm, and the anatase phase with a small percentage of rutile phase is the most suitable titanium dioxide phase composition for photocatalytic and superhydrophilic activities [33].
In 2013, Reza Asfipour et al. investigated the self-cleaning of wool fibers dyed with metal complex dyes and treated with nano titanium dioxide. In this research, wool fabric was dyed with metal complex dyes and it was completed with nano titanium dioxide in all processes in this project. Ultrasonic technology has been used, which is a clean method to disperse nanoparticles. For this purpose, the 100% wool fabric is first washed, and then oxidized with a solution containing potassium permanganate. After the oxidation process, the color of the wool fabric is changed by sodium bisulfite. It has been dyed, and then the dyeing and finishing solution including different percentages of titanium nanodioxide, metal complex dye, citric acid, and sodium hypophosphite was prepared, and the woolen product was processed by checking the results of EDX tests. SEM XRD proved the existence of titanium nanodioxide on the wool product [34].
Hamira Jamali and Hekaran investigated the effect of annealing temperature on the hydrophobic properties of moisture-insulating nanoparticles based on organosilane compounds in 2013. In recent years, a lot of research has been done to make artificial hydrophobic surfaces with different techniques to imitate natural self-cleaning surfaces so that the hydrophobic coatings are prepared based on organic, inorganic compounds, and organic-inorganic hybrids. In the present study, the organic-inorganic hybrid hydrophobic silica layer was prepared by sol-gel process and coating by immersion method. Sol solution during the hydrolysis and condensation reaction of tetramethoxysilane (TMOS) diluted in methanol in the presence of acetic acid as a catalyst and dimethyl octadecyl (3-(trimethoxysilyl)propylammonium chloride AEM) as a hydrophobic agent, at room temperature of 27 C prepared and simple immersion method was coated on the sodalime glass substrate. The hydrophobicity properties were investigated by changing the annealing temperature, and the highest hydrophobicity was obtained at 160 °C. The morphology of the prepared layer and chemical modification of TMOS surface were studied by atomic force microscope (AFM) and Fourier transform infrared spectrometer (FT-IR), respectively. Hydrophobic properties and thermal stability were investigated by measuring the water droplet contact angle and STA thermal analysis.
In 2012, Abbas Akbarpour et al. worked on the use of a combination of Tio2 and SiO2 nanoparticles to create self-cleaning concrete with high resistance. Concrete can be called one of the most widely used materials in building construction. According to the mentioned characteristics of concrete, special changes can be made in the properties of concrete by adding nanoparticles to it, one of the most used nanoparticles in concrete is Tio2, SiO2, each of which adds special properties to concrete. In this article, while examining the characteristics of these nanoparticles particle and also their effects on concrete, a plan for combined use in order to create self-cleaning concrete with high resistance has been presented [35].
In 2013, Maryam Kodkhodaei et al. worked on applying a self-healing coating based on titania-benzotriazole ceramic on the surface of aluminum 7075 using the sol-gel method. The main role of the coating in corrosion protection is to create a dense barrier against corrosive environments. However, the damage caused during the operation of coated structures allows direct access of corrosive agents to the metal surface. Nowadays, hybrid coatings have been used in industries to a significant extent due to their favorable mechanical properties, good resistance to scratches and corrosion. In this research, to improve the surface properties of aluminum 7075, titanium base self-healing coatings have been applied on the surface of aluminum 7075 by sol-gel method under immersion process. The type of links, structural properties, and morphology of aluminum 7075 have been investigated by FTIR and FESEM analyses. Likewise, the corrosion properties of the coating in 5.3% NaC1 solution have been evaluated by electrochemical methods such as Tafel polarization and impedance. The results show that the application of hybrid nanostructure coating is homogeneous, uniform, and crack-free, which has improved corrosion resistance [36].
In 2014, Gholamali Shafabakhsh et al. worked on the laboratory evaluation of fatigue of warm asphalt samples modified with TiO2 nanoparticles. In the past several years, bitumen modification with various additives has been studied to increase asphalt fatigue resistance. Nanoparticles and the new world of application of these particles have marked a new scientific revolution. These particles with extremely small sizes, very high surface activity with the lowest amount of consumption compared with the other bitumen additives, have made the asphalt mixture more resistant to environmental and traffic destructive factors. In this research, 2%, 4%, and 6% of TiO2 nanoparticles of Bahir 60-70 were combined and after performance tests, the samples made with bitumen modified with 4% of these nanoparticles were subjected to the fatigue loads of the 4-point bending beam test. The results have shown that titanium oxide nanoparticles have increased the fatigue life of asphalt by modifying the functional properties of bitumen and these samples have a higher breaking point [3].
In 2014, Fateme Tawanger et al. worked on improving the surface structure of woolen goods using the self-cleaning property of nano titanium dioxide. In this regard, woolen fabric with different percentages of 0.25, 0.5, 0.75, 1, and 1.25 of nano titanium dioxide and different amounts of citric acid (CA) (80 gr/l), 60, 40, and 20 were crosslinked by immersion method. The degree of self-cleaning of fabrics treated and stained with natural dyes such as coffee and black and synthetic dyes that include dispersed and direct dyes under the radiation of ultraviolet (UV) lamp 400 watts and sunlight, with it has been measured using reflectance spectrophotometry. Furthermore, the study of the structure and morphology of the coating of titanium nanoparticles on wool fabric was investigated with a scanning electron microscope (SEM) and the crystalline state of titanium nanoparticles was investigated by X-ray diffraction (XRD). The results show that with the increase in the percentage of nanodioxide titanium's self-cleaning property also increases, and also the amount of self-cleaning of oxidized fabrics treated with the crosslink method is higher than non-oxidized fabrics, and at the same time, the self-cleaning ability of fabrics treated under the radiation of a 400 watt UV lamp compared with sunlight, it is more. Given that the increase of citric acid in a fixed amount of nano-titanium dioxide has also been investigated, we came to the conclusion that the amount of citric acid is suitable up to an optimal level, and its more application will reduce the amount of self-cleaning [36].
Materials and methods
The centrifuge rotates the liquid sample at a very high speed using centrifugal force. There are different centrifuges depending on the sample size and capacity. The speed of centrifuges is measured according to the amount of centrifugal energy entering the sample, and it is usually measured in revolutions per minute (RPM). The sedimentation speed of the particles in the centrifuge depends on the size and shape of the particles, the acceleration of the centrifuge, the volume fraction of solids, the density difference between the particles and the liquid, and viscosity. Centrifuges usually have a rotor in which there are 2-4-6 or many wells in which special centrifuge tubes are placed. When a suspension is placed in a centrifuge, the solids settle at the bottom of the test tube and create a mixed wall. This wall allows the supernatant to be easily separated from the suspension. During the centrifuge, the door is locked. The closed door protects the person from hitting the rotor, which is rotating at a very high speed. The rotor housing and the rotor door are designed in such a way that if the pipes break even at the maximum speed, there is no danger to the user. Before the centrifuge, the tubes in the rotor should be balanced using a blank or other samples. The magnetic stirrer is one of the common laboratory tools that is of particular importance. This device is used to stir liquids by using a rotating magnetic field. This device consists of a plate on which the desired solution is placed in a beaker or any other suitable container. Under the screen there is a magnetic axis that can rotate. When the solution is placed on the screen, a small piece of magnet called a magnet is placed inside it. When the magnetic axis under the plate of the device starts to rotate, it moves the solution magnet with it, and the magnet starts to rotate in the solution, and thus the solution starts to rotate. One of the important benefits of the magnetic stirrer device, which has increased its use in laboratories, is that it causes the solution placed on the device to be stirred uniformly. In magnetic stirrers with a heater, there is a heating device under the plate that heats the plate. It heats up and causes the solution to be heated uniformly, and the temperature of the stirring plate can be adjusted according to one's desire by using the screw installed on the device.
The sol-gel process is not a new method. In 1800, Abelmann accidentally observed that silicon tetrachloride (SiCl4), left in a container, first hydrolyzed and then turned into a gel. In 1950, extensive studies on the synthesis of ceramics and glass structures using this method began. It is worth mentioning that with this method, many inorganic oxides such as TiO2, SiO2, ZrO2 were synthesized. Aerogels are one of the products of the sol-gel process. The simplest definition of an aerogel is a dry gel obtained by removing moisture from a wet gel. Depending on the method of moisture removal, the structure of this gel largely preserves the structure of the wet gel (initial gel). Wet gel can be prepared by different processes. These methods are generally referred to as sol-gel methods (Figure 1 & 2). Below is a summary of the chemical reactions involved in the sol-gel method:
In general, in any chemical process, the product obtained is the result of different chemical reactions. In the sol-gel process, various reactions occur to obtain the final product. In this part, we will discuss in detail the most important reactions that the sol-gel process is based on.
Sol-gel process steps
To produce a product by sol-gel method, it is necessary to first provide the prerequisites and conditions for the reactions.
Gel formation
For this, it is enough to stimulate the created solution in some way, so that the finely dispersed particles (each of which contains several to several tens of molecular or atomic units of the corresponding initiators) begin to assemble. By creating (physical and chemical) interactions between the suspended particles dispersed in the cell solution, they gather together as units consisting of several tens of thousands of molecules and form an infinitely large three-dimensional molecule, which usually takes up the entire volume of the container. It takes over the reaction. This giant molecule, which has many pores, traps all the solvent inside, and is called a wet gel.
To produce a wet gel, we need to stimulate the sol solution to reach the gel. This stimulation can be done using a suitable reagent (pure water or water with NaOH, HCl). In fact, in the step of converting sol to gel, which is controlled by reactions called condensation, it is considered an inorganic polymerization reaction, the final product of which is an oxide network containing M-O-M metal oxide clusters.
The condensation reaction is exactly the opposite of the hydrolysis reaction. In hydrolysis, coarse molecules are converted into simpler components by consuming water, but in condensation, two simple molecules join together and form a more complex molecule. At the edge of this combination, a small molecule like water is released. A condensation reaction occurs when two metal hydroxides (or one hydroxide + one alkoxide) (M-OR+HO-M) combine to form a metal oxide (M-O-M). The gel is finally made as a solution. In the next step, we have to separate the solvent from it in a way to complete the process. There are different types of gels, each of which has its own characteristics and uses. Depending on the type of solvent used or the type of drying method, their names, properties and applications will be different.
Sol gel and its products
The sol-gel process is a bottom-up synthesis method. In this process, the product of a number of chemical reactions is irreversible. In fact, these reactions cause the transformation of molecules of the initial homogeneous solution as sol, into an unlimited, heavy and three-dimensional polymer molecule as gel. As an example, the hydrolysis reaction followed by the condensation reaction.
Binary or ternary systems can be produced by mixing different primary salts. Each of the primary salts has its own reaction speed, which depends on conditions such as pH, concentration, solvent and temperature. The created polymer gel is formed in the form of a three-dimensional skeleton, which causes the holes to connect, and after drying, it shrinks and creates a solid solid. It can be mentioned that the final materials and products can be designed in such a way, which will cause the emergence of nano-pores, which will result in a much higher specific surface area.
In the sol-gel process, the transformation of the sol into the gel state is often achieved by changing the pH or changing the concentration of the solution. The main reasons for using the sol-gel process are to produce a product with high purity, narrow particle size distribution and achieve a uniform nanostructure at low temperature. The sol-gel method is often used for the synthesis of metal oxides.
The sol-gel process involves changing the state of sol to gel using different techniques, most of which use slow and gentle drying to remove the solvent. It should be noted that due to the phenomenon of shrinkage during the drying of the gel, during the drying process, the necessary points should be observed to prevent the occurrence of cracks. The produced gel can be casted, and by using it, molded parts can be made and by drying them, a single piece can be obtained. The pieces obtained in this way can be used as filter or membrane.
It is also possible to produce thin films with a thickness of 50-500 nm on a substrate by performing Spin Coating and Thin Films. These produced thin films have wide applications in terms of electronics, wear, or chemical applications, and in addition, they can have an effect on optical properties.
Continuous pores on the nanoscale can be used as a place to load with secondary materials [4]. This process is done using methods such as purification in the molten phase or chemical reactions. Such materials are classified in the category of nanocomposites. One of the useful uses of these porous materials is that they fill these voids with various industrial catalysts, and due to the very high active level of these porous materials, the catalytic efficiency is greatly increased and reduces the total price of the manufactured product.
Drying
The characteristic of gels is that they can take the shape of the container in which they are produced. Depending on the strength of the gel structure, an independent piece with large holes can be obtained. Because in this situation, the solvent is one of the internal components of the gel (it keeps the network). It is very difficult to maintain the stability of the pore structure by removing the solvent. It is usually necessary to allow the gel structure to mature before drying so that the bonds between the particles become stronger. This stage lasts from several hours to several days. This process is called aging. During the aging process, the gel continues to change so that new bonds are formed and the strength of the gel skeleton increases.
In the sol-gel process, in the initial stage, chemicals form nanometer particles called sol in the solution. In the next step, these particles join together to form a solid three-dimensional network called a gel. After the aging process and preparing the gel, they are separated. After preparing the gel, the liquid phase (solvent) is separated in different ways.
1- Leave the material in the environment to dry by itself. In this case, the resulting material is called Xerogel and has fewer holes and is dense. Xerogel refers to a gel in which all the liquid inside the gel cavities has been removed, so that the structure has become slightly denser and more compact, and the wrinkling compared to the state of the wet gel is quite evident in it.
2- Another way is the supercritical method, in which the changes of the solid network are minimized. The resulting material has a porous network, which is called an Aerogel. Aerogel is also a type of dry gel. Therefore, the solvent has been removed from the gel. Here, the solvent exit was done in such a way that there was no contraction or change in the gel structure. This type of gel is generally prepared by heating the gel to the critical temperature of the solvent. Therefore, there is no equilibrium between the liquid and the vapor, and a gel with a high specific surface area is obtained, while maintaining the structure, compared with the wet gel state. Unlike zerogel, in aerogel all pores and structure are preserved and not condensed.
Preparation of transparent and thin film
Based on Table 1, we change the amounts of epoxy silane and distilled water plus isopropanol and form a sticky film and pour it into the glass and note the drying time. We choose as the ideal movie.
Discussion
25 grams of titanium sulfate was added to 50 cc of distilled water, and then ammonia was added dropwise under the hood until it becomes a jelly. Thereafter, a filter paper was placed in a funnel, the jelly solution was poured, and passed through the filter solution, and then deposited . In this case, white sediments was remained on the filter paper. Next, the sediments were washed with distilled water to remove the ammonia. After that, the sediment was poured into a beaker and add distilled water to it (Figure 3). Then, a small amount of hydrogen peroxide was added to it. It could be seen that the mixture turns yellow little by little. This mixture was put on a magnetic stirrer for 1 hour until it turned into a gel. The gel was put in a centrifuge, and then it was passed through the filter paper and washed with distilled water, and then the gel was dissolved in distilled water for 2 hours at 100 C.
Today, due to the industrialization of human societies and the need to use fossil fuels as much as possible, the face of most cities in the world has become black and dirty due to pollution and fats from these fuels. In proportion to the progress of societies, the discovery of nano-sized materials can be the solution to many problems. Among them, two types of self-cleaning and hydrophobic coatings have been developed, which are based on hydrophobic properties, but with the difference that self-cleaning coatings are possible to clean surfaces only by sunlight [4-6]. By creating a coating of this material on the exterior of buildings and various structures, it is possible to break down existing pollution, which are mostly hydrocarbon compounds, only by using sunlight, and by using only water, these pollutions can be cleaned. This property can be useful to keep all the internal surfaces clean and new, which will prolong the life of various colors and building structures. In addition to creating anti-water properties, nano self-cleaning solutions are very effective with the use of statin light that removes dust from surfaces [10]. Due to their special optical and electrical performance, nanoparticles have a wide range of applications in many fields of diagnostic and therapeutic tools. Titanium is one of the widely used metals and is the ninth most abundant element on the earth's crust, which can enter the food chain as a bleaching agent in dairy products, chocolate, milk powder, soy products, sausage, and the other materials. It is estimated that about 300 mg of titanium is used daily in food. Titanium dioxide nanoparticles are currently used industrially in many consumer products such as paints, food, cosmetics, toothpaste and environmental disinfectants. It is also used in tumor treatment, drug delivery, and gene transfer to cells and tissues. There is a lot of evidence about the potential risks of titanium dioxide nanoparticles on health and the environment due to their physicochemical properties, including their shape, size, surface characteristics, and internal structure. These particles enter the body in different ways, including inhalation, skin penetration, and ingestion. In the digestive system, it is absorbed by the lymphatic tissue of the intestine and enters the blood vessel and is easily distributed throughout the body and may penetrate the cell membrane and enter the mitochondria and disrupt the mitochondrial energy pathways including oxidative phosphorylation and glycolysis and as a result The reactive oxygen species created cause oxidative damage in the body.
Conclusion
Among the mentioned cases, the most research has been done on the nanocatalyst activity. All investigations have been done to increase the catalytic activity of nanocatalysts. Drying is possible by a simple evaporation process, or by removing the solvent under vacuum. As a result of the capillary forces created by the interaction between the solvent and the walls of the material structure due to the solvent evaporation, the porosity is expected to decrease. If so, this change is irreversible after the gel dries. One way to deal with this problem is to eliminate the contact surface between the gas and liquid phases in the system. This issue can be achieved by removing the solvents when they are in their supercritical state. The supercritical point in the phase diagram is where gas and liquid do not exist as separate phases.
Acknowledgements
We would like to thank all the people who helped in preparing and compiling the article and collecting the available data.
Conflict of Interest
There are no conflicts of interests.
Orcid:
Seyedeh Shahrzad Moayeripour: https://www.orcid.org/0000-0002-0000-0000
--------------------------------------------------------------------------------------
--------------------------------------------------------------------------------------
Copyright © 2023 by SPC (Sami Publishing Company) + is an open access article distributed under the Creative Commons Attribution License(CC BY) license (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

.png)
.png)
.png)
.png)
