Document Type : Review Article
Authors
- Nafise Nikkerdar 1
- Amin Golshah 2
- Mohammad Salmani Mobarakeh 3
- Nima Fallahnia 4
- Bahram Azizi 5
- Ehsan Shoohanizad 5
- Razieh Souri 3
- Mohsen Safaei 3, 6
1 Department of Oral and Maxillofacial Radiology, School of Dentistry, Kermanshah University of Medical Sciences, Kermanshah, Iran
2 Department of Orthodontics, School of Dentistry, Kermanshah University of Medical Sciences, Kermanshah, Iran
3 Advanced Dental Sciences and Technology Research Center, School of Dentistry, Kermanshah University of Medical Sciences, Kermanshah, Iran
4 Students Research Committee, Kermanshah University of Medical Sciences, Kermanshah, Iran
5 Department of Oral and Maxillofacial Surgery, School of Dentistry, Kermanshah University of Medical Sciences, Kermanshah, Iran
6 Division of Dental Biomaterials, School of Dentistry, Kermanshah University of Medical Sciences, Kermanshah, Iran
Abstract
Numerous compounds with a variety of properties are used in dentistry for diverse purposes. Alumina, zirconia, lithium disilicate, silica, and zirconia are the finest materials for dental work. For several reasons, zirconia-based materials are now one of the most challenging research topics. Zirconia, sometimes called ceramic steel or zirconium oxide, is used in dentistry owing to its excellent properties, robust resistance to erosive forces, low cost, high biocompatibility, and leathery texture. In addition, the mechanical properties of Zr-based bulk metal complexes are essential for usage as dental implant materials. Over the last several years, zirconia nanoparticles (ZrNp) have made remarkable advancements in dentistry. ZrNp may significantly enhance the bionic and mechanical characteristics of dental ceramics and tissue engineering scaffolds. This review examined various aspects of ZrO2 particles and their applications in dentistry and medicine.
Graphical Abstract
Keywords
Main Subjects
Introduction
Zirconia is widely recognized in the ceramics industry for its remarkable characteristics of hardness and ability to withstand fractures under normal conditions. In addition, the fine grain size of the material, which is less than a micron, allows for the achievement of outstanding surface finishes and the capability to maintain a sharp edge. Researchers and manufacturers have developed complex formulations to prevent the spread of cracks, with yttria-stabilized tetragonal zirconia polycrystal (Y-TZP; zirconia) being a common ingredient [1,2]. The advent of zirconia ceramics, which co-occurred with electronic technology, has allowed dental research and business to realize their goals. Zirconia has gained popularity in biomedical applications, particularly surgical implants, due to its aesthetic properties and biocompatibility. It is also widely used in dentistry, including for crowns, bridges, implants, and veneers; this material is very biocompatible and can withstand the long-term effects of the oral cavity's thermal, chemical, and mechanical stresses. There has been a "Big Bang" in the dental industry in the last ten years when it comes to manufacturing zirconia for various dental uses [3,4]. The previous developments were identified by a worldwide advancement creating significant assumptions. However, this novel technology requires a particular mass of time entirely confirmed by dentists and dentists. The dental profession has little clinical information concerning intensity resistance under bonding effectiveness, color performance, longevity, and fatigue of the zirconia-oriented reconstruction [5].
In previous decades, metal-ceramic restorations have been considered the benchmark in the field of fixed prosthetics within the dentistry realm. Zirconium-ceramic crowns provide a viable alternative to their metal-ceramic counterparts. Now zirconia technology has advanced to the point that it can be used in CAD/CAM applications, it can improve everyday dentistry [6]. A machine and the manufacturer's essential software (CAD) are required for the three-dimensional design of Y-TZP frameworks. After a scanning technique, the data are transmitted to the computerized manufacturing unit (CAM), which presents the preset formation of the zirconia system [7]. Zirconia-based frameworks are created by milling out from a hard block (subtractive technique), mainly for Y-TZP ceramics, or through electrophoretic deposition (additive method), especially for cerium-tetragonal polycrystal (Ce-TZP) ceramics [8]. Zirconia fragment milling can be done as described in section [9] or, if required, in stage 10 using suitable cutting diamonds and water coolant. Milling partially sintered Y-TZP ceramics using dry carbide burs is possible, which is why many CAD/CAM systems employ this material. The anticipated milling size (partially sintered frame) is roughly 20% to 25% larger than the primary dimensions during the design phase in shrinkage anticipation during the final sintering process [10].
Furthermore, milling full sintered or hot isostatic press (HIP) zirconia blocks takes time because of the stone’s enhanced stiffness. Still, there are no dimensional alterations (i.e. shrinkage). The processing of partially sintered Y-TZP ceramics at room temperature depends on the in depth destruction or surface (i.e. voids, defects, and cracks) [11], as opposed to heavy completely sintered (or HIP) machining, which may cause microcracks [12].
Ceramics find utility across diverse applications, such as the chemical industry, electronics, and biomedical engineering, attributable to their diverse and superior features. These commendable attributes encompass exceptional mechanical robustness and resilience, favorable thermal and chemical steadfastness, as well as feasible thermal, optical, electrical, and magnetic functionality [13]. Fixed partial dentures cannot be utilized because of traits like fragility. As a solid and novel ceramic material, zirconium oxide (ZrO2: zirconia) has been progressively considered in dentistry since the turn of the century [14]. Initial implementation relied on fixed partial dentures that contained no metal. Despite this, it finds use in a wide variety of dental restoration and prosthetic contexts, including orthodontic braces [15], fixed partial posterior dentures [16], and abutments or fixtures for dental implants [17]. Thus, zirconia-based ceramics are complete permanent dental prostheses that last a long time and do not include metal [18].
Medical and dental applications
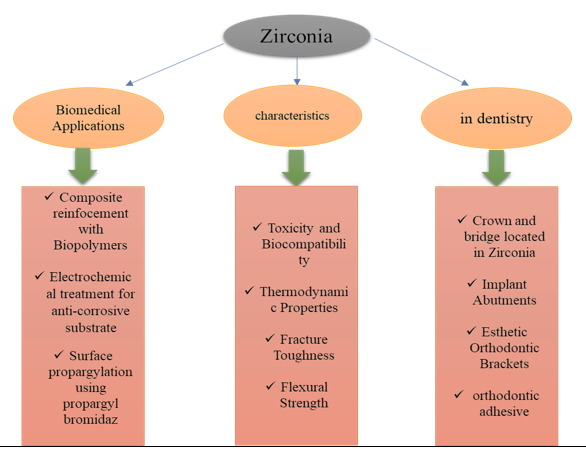
The use of zirconia in dentistry and medicine has rapidly increased over the last decade due to its advantageous physical, esthetic, biological, and corrosion characteristics. Zirconia orthopedic hip replacements have shown superior wear tolerance to most systems. However, the possibility of traumatic rupture remains problematic. Furthermore, zirconia biomaterials are synthetic bone fillers to heal bone defects. Zirconia has other clinical applications, such as arthroplasty. Zirconia has orthopedic applications, including knee and hip prostheses, strictly supporting hip joint heads, dental crowns, and tibial plates [19]. Some of the most important biomedical applications of zirconia are displayed in Figure 1.
Ceramics are increasingly advanced as high-strength dental prosthesis products [20-22]. Modern high-speed sintering technology [24,25] and chair-side milling [23] have made the automated, dependable, and time-efficient manufacture of dental restorations possible. Increasing the durability and aesthetic quality of ceramics is the primary objective of the goods. Prosthetic dentistry is a profitable and vast health sector, but small advances in products and processing technology may have enormous economic implications for patients and providers. Y-TZP is the most potent of all remedial ceramics. Different variants of Y-TZP exist based on additives and dopants, corresponding heat treatments, and sintering profiles [26,27]. The main draws are their biocompatibility, corrosion resistance, and outstanding mechanical capabilities. The biggest challenge is making them seem good enough to pass for the same type of teeth. Y-TZP, especially silicates derived from lithia, can hold their own against weaker but more translucent glass ceramics [28]. When aesthetics failed, the conventional approach used a powder-fired porcelain set on a zirconia core [29]. The residual thermally induced tensions make chipping and delamination possible even in this bilayer structure. Some have tried to mitigate this risk by machining the veneer and structure separately, and then fusing them using resin luting factors (such as VITA Rapid Layer Technology or Ivoclar IPS e.max CAD-on Technology). Grinding agents are compatible but won't eliminate residual tensions by making the veneer easier to flange and fuse. More tooth structures will need to be removed to accommodate veneers because they increase reconstructive thickness. Therefore, the most recent developments in monolithic drive restorations have focused on reducing material thickness requirements, improving manufacturing transparency, and balancing cosmetic requirements with consistency. Because Y-TZP and other ceramics do not increase the strength of metals, any flaw in the prosthetic's construction can cause fractures to manifest. Long-term defeats from a range of forms of fracturing are a persistent concern [30]. Some of the most critical applications of zirconia in dentistry are depicted in Figure 2.
Classifications
Three different kinds of t′-zirconia materials are now being investigated for dental applications. t-ZrO2 single-phase includes t-ZrO2 two-phase materials as the trivial phase (dispersed and precipitated, respectively), and the final phase [31] t-fixation occurs when three substances are combined. Toughness is signified by the martensitic transformation t → m:
- Ceramics that have been hardened with zirconia, often known as zirconia-toughened ceramics or, more commonly, dispersion-toughened ceramics, are: Extremely embedding elastic modulus changing zirconia grains form a matrix; two examples of this type of material are ZrO2-toughened alumina (Al2O3; ZTA) and ZrO2-toughened mullite (3Al2O3・2SiO2; ZTM) [32,33].
- The tetragonal step precipitates in the matrix of partially stabilized zirconia (PSZ), which consists of a matrix of cubic zirconia infixing transformable t-zirconia grains;
A material entirely made of transformable t′-zirconia grains is known as tetragonal zirconia polycrystals (TZP). Yttrium oxide-stabilized TZP, abbreviated as Y-TZP or 3Y-TZP, is the mainstay of dense-sintered zirconia's current dental applications [34]. The Y2O3 required for complete cubic stabilization is eight mol% [35]. Depending on particle size, tetragonal zirconia can achieve partial stabilization at a concentration of 2–5 mol% (often three mol% is used). The reasons behind TZP's spread instead of ZTC and PSZ are evident. PSZ is challenging, and ZTCs have fewer mechanical features than TZP and PSZ. Several factors influence the precipitation stage of processing, including the first-grain powder scale, the stabilizer and material shape, the time and temperature required for nucleation, and the formation of tetragonal precipitates [36]. The various crystal structures of zirconia are demonstrated in Figure 3.
Zirconia application in biomedical
In dentistry and medicine, load-bearing surfaces are commonly used as dental and medical prosthetics, such as crowns/bridges and joints, which are increasingly required by millions of citizens in our aging populations. Various products, including ceramics, plastics, polymers, and composites, are used for these functional surfaces. Among these elements, zirconia mixes an extreme power and strength of fracture, low thermal conductivity, slow crack growth resistance, high ionic conductivity, chemical inertness, and attractive biocompatibility [37-39]. For a long time, zirconia has been utilized in ferrule engineering of optical fiber connectors, mechanical components, and environmental filters. Zirconia has been used for total hip replacements in surgical implants (such as femoral heads) for nearly fifty years [40]. For the last twenty years, it has been utilized in restorative dentistry [41,42] as dental abutments, implants, and cores for bi-layered posteriors, including dental bridges and crowns. Using newly colored zirconia with demonstrated translucency allows accurate color matching to human teeth. The breaking strength of this modern material can reach 6 MPa m1/2/2, and its flexural pressure ranges from 900 to 1400 MPa [43]. Zirconia bridges and monolithic crowns for back functions have significantly increased the prevalence of these benefits [44]. When used in dentistry and medical equipment, zirconia can perform mechanical tasks like wear and tear resistance and biological capabilities like bacterial decolonization, bonding strength, and cell adhesion. These capabilities are achieved through surface treatment and machining of the material. Synthetic bone fillers made of zirconia can fix broken bones [45]. Arthroplasty is one of its further medical applications. Hip joint heads often include dental crowns, tibial plates, temporary braces, and zirconia and yttria-supported zirconia, which serve orthopedic purposes (e.g., in knee and hip prostheses) [46]. Several zirconia systems are available, but only three have found dental use. First, there is zirconia-doped alumina (ZTA); second, there is magnesium cation-doped zirconia (Mg-PSZ); and third, there is yttrium cation-doped tetragonal zirconia polycrystal (3Y-TZP) [47]. Even though TZP and PSZ are suitable for use in the medical field, the presence of zirconia toughened alumina (ZTA) bacteria altered the effectiveness of joint replacements and led to long-lasting infections. The accumulation of bacterial plaques on dental restorations can exacerbate dental caries, periodontal issues, and inflammation of the gums [48]. The roughness and surface energy of restorative chemicals are typically connected to plaque preservation. Rougher textures promote bacterial colonization, according to medical research. The fissures of vital teeth causing symptoms are a typical place for bacteria to hide [49].
A surface roughness of 200 nm Ra has been determined to be necessary for the preservation of bacterial plaques in vivo investigations of various materials, including gold, amalgam, human enamel, composite resin, ionomer glass, acrylic resin, and porcelain, after undergoing multiple surface treatments such as polishing, condensing, scaling, scraping, finishing, or glazing [50]. Since all zirconia implant surfaces (roughness: 119-259 nm Ra) have been documented with specific bacterial adhesion [51], it is unknown whether this threshold should be applied to zirconia surfaces. Zirconia has several common medical uses, as presented in Table 1.
Dentally relevant zirconia characteristics
Toxicity and biocompatibility
In addition to be very biocompatible, zirconia is entirely non-toxic. No toxicity signs or adverse effects, no cytotoxicity on cytosomes, and no rejection response following implantation are characteristics of the hydroxyapatite-zirconia composite (HA-ZrO2), one of the most applied and considered ZTC orthopedic substances [62,63]. It also has excellent bone adherence, cell proliferation and metabolism, and a reasonable hemolytic rate. Superior osteoinductive potential in rabbits has also been demonstrated by graded HA-ZrO2 [64]. Compared to pure ceramic oxides, ZrO2-TiO2 based on ZTC has shown fast cell growth, extreme biocompatibility, and hardness generation proportional to the ZrO2 content, with slightly higher cell growth than TiO2 and ZrO2 tests [65]. Gingival fibroblasts (HGF) are not cytotoxic to Y-PSZ. A bioactive glass coating can further enhance the in vitro bioactivity of Mg-PSZ. Y-TZP's biocompatibility, mutagenic and carcinogenic effects have been successfully examined, and [66,67]. Enhancing the bioactivity of ZrO2 and the answer to osteoblast-like cells can be achieved by coating it with calcium phosphate and phosphate-based glass. When contrasted with titanium implants, ZrO2 implants demonstrated comparable levels of biocompatibility and osseointegration. There appears to be no effect on GF adherence and proliferation from coloring the zirconia with metal dopants [68].
Thermodynamic properties of glass and its transition temperature
The residual stress that arises is an outcome of the dissimilarity in the coefficient of thermal expansion (CTE) between the veneering ceramic and zirconia. This dissimilarity is likely to instigate a zirconia phase transformation, which subsequently impacts the bond strength. The coefficient of thermal expansion (CTE) measures the relative size change at a constant strain as a function of temperature. With pressure remaining constant, it describes how an object's size varies in response to changes in temperature. Tg is the temperature at which glass viscosity reaches 1012 Paმs, according to the International Union of Pure and Applied Chemistry (IUPAC) [69]. "The near mid-point of the temperature spectrum across which the glass transforms between elastic and viscoelastic activity has been calculated by a rapid shift in its coefficient of thermal expansion" [70], according to the International Organization for Standardization (ISO), provides a more understandable explanation. The CTE and Tg of veneering ceramics and ZrO2 are different because of their chemical diversity. Upon heating to the firing temperature, the zirconia core/framework and veneering ceramic exhibit thermal expansion by their respective specific thermal expansion coefficients (CTEs). The veneering material would be in a viscous-liquid state, and distinct sliding and adjustment on the solid zirconia would be possible if the ceramic temperature were higher than Tg. Volumetric compression occurs to the materials as they cool. The thermal contraction causes the material to change from a liquid to a solid when the temperature drops below the ceramic's glass transition temperature (Tg). Residual thermal stress (RTS) can occur during this technique due to thermal mismatch between the core material and the veneering ceramic. The strength of the bond between zirconia and ceramic is partially attributed to the compressive force generated as a result of the suitable difference in the coefficients of thermal expansion (CTE) between the veneering ceramic and zirconia. However, the excessive residual stresses that arise from a mismatch in CTE values can compromise the bond strength [71-73]. A veneering ceramic with CTE near the zirconia core/framework can help reduce RTS. The importance of CTE compatibility between the core and veneering materials was determined to be ±1 ppm/°K, assessed from 25 to 500 °C, based on a limited component analysis study [74]. It was thought that a far higher number of clinical failures may happen than this number. A reasonable degree of RTS is likely to occur at all times due to the difficulty in creating a veneering ceramic with zirconia-level CTE. The cooling rate [75-77] and the number of firing phases [78] play crucial roles.
Fracture toughness
For extended loading rates in mostly linear-elastic conditions with little plastic zone change, KIc is defined by the American Society for Testing and Materials as a crack extension deficiency under a crack-tip plane strain in Mode I [79]. Whatever the stresses applied or the shape of the cracked body, the following global shape is maintained for stress close to the crack tip in linearly elastic solids:
It is assumed that the loads and the geometry of the fracture line are symmetrical, and σ is the stress. KI is the "stress" intensity element dependent on the load and crack geometry. r and θ are polar harmonizes focused at the top of the crack. KI is at the crack edge, which indicates a high level of stress.
From this analysis, Irwin [80] derived a fracture criterion, stating that crack development occurs when the stress strength component reaches a critical value, KI = KIc, where KIc is the fracture power, an experimentally determined material constant ("I" for Mode I and "c" for critical"). There are a variety of proposed tests for determining ceramic fracture toughness:
- Single Edge V-Notch Beam (SEVNB, ISO 6872) [81],
- Single-Edge Precracked Beam (SEPB, ISO 15732),
- Surface Crack in Flexure (SCF, ISO 18756) [82], and
- Chevron-Notched Beam (CNB, ISO 24370) [83].
The ISO recommends the SEVNB technique for dental ceramics. According to ISO, this approach "has undergone international estimation for standardization and was discovered to be user-friendly, easy, accurate, and reliable" [84]. Using surface crack lengths linked to the angles of Vickers indentations to determine fracture toughness is discouraged by ISO [84] and is an example of a method that relies on notch crack lengths. Finding the "real" KIc value requires that the notch root's radius be proportional to the primary microstructural feature size. Otherwise, the computed gain is inflated, as demonstrated by the authors [85]. It is difficult to compare the fracture toughness of dental ZrO2 due to the aforementioned reasons.
Flexural strength
The flexural strength, denoted as σ, is the capacity of a material to resist bending and is often expressed in megapascals (MPa = N/mm2) for both solid and delicate materials [86].
So far, the only ceramic that can be classified as Class 6 and used as a substructure ceramic with four or more units is Y-TZP. When it comes to dental ceramics, three flexural test procedures work well [86]:
- Three-Point Bending Test (3PBT),
- Four-Point Bending Test (4PBT), and
- Biaxial Flexure Test (BFT).
A bar-shaped rectangular specimen with a width of 4 mm and a thickness ranging from 1.2 to 3.0 mm is used in 3PBT and 4PBT. One or two support rollers hold the specimen in position; for 3PBT, the lowest span is 12 mm, and for 4PBT, it is 16 mm. The specimen used in BFT is disc-shaped, with a diameter of 12-16 mm and a thickness of 1.2 mm. It is supported by three balls spaced at 120° angles and is loaded using a flat punch. Up until the point of failure, every experiment employs a static load [86]. While calculating the flexural strength of CAD/CAM materials is necessary, there is an additional practical benefit to using 3PBT and 4PBT instead of BFT. Most CAD/CAM materials are sold in pre-made blocks with specified dimensions. Compared to BFT, which requires generating a disc, producing the rectangular-section bars necessary for 3PBT and 4PBT is far more accessible. Manual trimming is needed to obtain a disc specimen with the required dimensions of 12-16 mm in diameter and 1.2 ± 0.2 mm in thickness, as drawing or milling a cylinder is not an option in most CAD/CAM software products. Surface grinding [89,90], air-particle abrasion [91], edge damage [87,88], and polishing are all known to cause zirconia specimens to be delicate. Previous research has shown that the results obtained from 4PBT (σ4PBT) are typically lower than those from 3PBT (σ3PBT) and BFT (σBFT) [92,93].
Color
Pure zirconia is typically an ivory or white hue. Industrial manufacturers study several coloring procedures (such as injection molding and heterogeneous nucleation) to generate a wide range of colors for t- and c-zirconia, which are used in optical components, structural units, ornamental materials, and ornaments [94,95]. Dental care is not the place for these methods. Three main coloring methods are now available to get shaded zirconia for dental use: First, in the industrial phase of production, using ZrO2 powder as a starting point, alloying with metal oxides yields pre-colored material. Second, special coloring liquids are used in the dental laboratory to infiltrate the green-phase zirconia. Finally, zirconia can be painted with liners after sintering and firing in a typical dental ceramic furnace. Among the many causes of delamination between porcelain zirconia restorations, the last approach has been considered a weak spot [96]. You can change the amount and mix of zirconia color characteristics with the help of metal oxides like Fe2O3, CeO2, Er2O3, Pr6O11, Bi2O3, and MnO2 [97,98]. It appears that mechanical properties are reduced more by Er2O3 than CeO2 [99]. According to the literature, the concentration, rather than the type of coloring material, affects the mechanical properties of ZrO2 [100]. Hardness and flexural strength are adversely affected by increasing immersion duration in green-phase color infiltration [101]. Changing a material's color without affecting its mechanical properties or crystal stage development is possible when the concentration of coloring metal oxides is kept low [102,103].
Translucency
The amount of light passing through a turbid material or reflecting off a substrate surface is one measure of its transparency [104]. Therefore, whereas a "transparent" material would allow sufficient light transmission to enable the perception of separate images, a "translucent" medium would create diffusion necessary to prevent the perception of distinct images. Glasses and viewports subjected to extremely high temperatures, color filters used in harsh settings, and electronic displays resistant to scratches and impacts are just a few of the many uses for transparent zirconia [105]. In addition to zirconia's transparency, the chemical makeup, number of crystals, and scale of particles about light wavelength all contribute to its overall appearance [106]. Pore spreading occurs when a pore size is more significant than 50 nm and is a substantial factor in transmitting light reduction [107,108]. Because it is implausible that the pore diameter will exceed the grain scale, the problem of light dispersion of zirconia is avoided in high-density nanocrystalline zirconia by using zirconia grains that are less than 50 nm in size [105]. When determining the sintered density, the sintering temperature is frequently crucial. Sintering zirconia crystals at higher temperatures produces a more compacted structure with less porosity, defects, and weaknesses [108]. Translucency is key in matching the teeth's natural shade [109]. The translucency parameter (TP) or contrast ratio (CR) are common metrics for this in the biomedical field [110]. Compared to other ceramics, ZrO2 is the least see-through, and research suggests that its thickness makes it clinically opaque [111-113]; the current description of zirconia as a "semi-translucent" core material is supported by the fact that, depending on its thickness and microstructure, zirconia can be seen as transparent up to a certain extent [114,115]. The translucency of shaded zirconia has been brought up by a few writers who have put forth the following theories: (i) the shadow effect does not clinically impact transparency, providing a background that matches the veneering porcelain's shade, and (ii) colored models exhibit less color variation when compared to noncolored samples or human dentine, rather than noncolored samples [104,116]. Whether you use heat pressing or typical condensing veneering ceramic, the veneering technique can impact the translucency of a zirconia-ceramic repair [117].
Zirconia: The essence of an imminent bioceramic discovery
Zirconium minerals have been used for decades and are still considered hyacinth jacinth and jargon. Zirconium alloy derives from the zargon, an Arabic word (In color, golden); in turn, Zirconium minerals have been used for decades and are still referred to as hyacinth jacinth and jargon. Zirconium alloy is derived from zargon, an Arabic word (in color, golden) originating from two words in Persian: zar (gold) and gun (color). Zirconia, an alloy of silicon and aluminum, was discovered in 1789 by German scientist Martin Heinrich Klaproth and was isolated in 1824 by Swedish scientist Jons Jakob Berzelius. Currently, zirconia is widely employed for industrial applications. The late sixties of the past century have shown examination and improvement of zirconia as biomaterial, while Helmer and Driskell revealed the first article on various zirconia biomedical functions [118]. The white crystalline oxide of zirconium is ZrO2, classified as zirconia. While there is no pure ZrO2, it can be detected in zirconia (ZrSiO4) and minerals such as baddeleyite. It is characterized by a hexagonal close-packed crystalline construction at ordinary temperatures and forms several combinations, such as the zirconyl and zirconate salts. Zirconia can be formed as a white powder with both acidic and base characteristics. There are three distinct stages in the crystallographic separation of zirconium dioxide crystals into crystalline cells (mesh): (i) A straight prism with square sides is called a cubic prism; (ii) A straight prism with rectangular sides is called a tetragonal prism; and (iii) A twisted prism with parallel sections is called a monoclinic prism. The cubic phase has modest mechanical properties and is continuous over 2.370 °C. Ceramics with enhanced mechanical characteristics can be obtained during the tetragonal stage, which occurs between 1,170 and 2,370 °C, and during the monoclinic phase, which remains at room temperature up to 1,170 °C, mechanical work is reduced, and ceramic particle adhesion is reduced as well [119,120].
Zirconia dental application
While several zirconia-based ceramic systems are now usable, only three are currently applied in dentistry. These are 3Y-TZP, ZTA, and Mg-PSZ [121]. Some common dental applications of zirconia are indicated in Table 2.
Dental posts focused on zirconia
The need for extra esthetic posts, except for all-ceramic restorations, has improved new post components. Metal posts can trigger negative esthetic effects, such as a gray discoloration of translucent all-ceramic crowns and the enveloping gingival margin, under conditions where all-ceramic restorations are used to repair anterior teeth [132]. In addition, caustic reactions to prefabricated posts can contribute to complexity involving metallic taste, sensitization, oral burning, oral discomfort, and additional reactions involving the enveloping tissues and oral surroundings [133]. These problems have contributed to strengthening white or transparent posts constructed of zirconia and other ceramic components. Various investigators have presented Steady zirconia ceramic to develop post-systems [134] because they possess excessive severity and toughness of being broken rather than other ceramics. Smooth, conical, and parallel or tapering zirconia posts at the crown and on the wreath side are accessible. They are rounded at the highest zenith to decrease stress concentration at the root crown. Other distinct include polyesters with sixty-five percent zirconium fibers, lower Young's module, and hardness than pure zirconia; however, the advantageous light transmission characteristics without adjustment [135]. Zirconia posts that can be used directly and indirectly are highly radiopaque and biocompatible and exhibit a strong light transmission by both root and coronal recovery [136].
Paul and Werder [114] also examined zirconia posts after 4.7 years of extensive clinical service and achieved significant clinical success with zirconia posts with explicit composite cores. Using in vitro samples, Kwiatkowski and Geller investigated the mechanical properties of zirconia posts [137]. According to their study, zirconia posts outperform all ceramic posts and cores in strength. Zirconia posts often deliver perceived benefits regarding esthetics and biocompatibility but have some limitations. Without any ductility, they are inflexible; hence, problems can be identified if they are limited and retreatments are necessary [138]. This means that zirconia posts lack highly reactive bonding abilities with radicular dentine during dynamic loading and thermocycling [139]. In addition, the ceramic posts have a greater retention effectiveness than the serrated posts [140].
Crown and bridge located in zirconia
The production of zirconia frames for crown and bridge of either pre-sintered or highly isostatic compressed zirconia has also been carried out [141,142]. Zirconia structures offer a fresh perspective for metal-free single-tooth reconstructions and fixed partial dentures owing to zirconium's intense flexural strength of over 900 MPa and exhibited strong initial clinical outcomes [143]. Tinschert et al. [144] compared the survival of many non-metal core prosthesis units and identified zirconium ceramics' apical beginning and longest-term strength with alumina oxide. Sailer et al. [145] clinically checked 58 zirconia bridges developed by the direct ceramic machining method. The findings revealed a retention rate of 84% for three and a half years. Minor chipping occurred in 11% of the bridges surveyed in the study. Tinschert et al. [146] built 65 zirconia bridges utilizing the DCS President® process. For three years, he and his colleagues identified the zirconia bridges. He described some chipping of the veneers in 6% of the bridges, indicating a total survival level of 86% [147].
Implant abutments based on zirconia
With zirconia ceramics for the construction, clinicians were comforted to continue their implantation-supported rehabilitation functions. As an implant-supported restoration, zirconia results from a greater toughness and less elastic modulus value. In stabilized and toughened structures, zirconia offers several superiorities over alumina to react to fragility complications and possible implant defeat [148]. Their matching tooth color, high tissue compatibility, and reduction in plaque accumulation characterize these abutments [149]. A comparative analysis of 30 zirconias vs. 51 aluminas has been performed by Yildirim et al. [150]. Accumulative survival rates of 100% and 98.1% were detected for every category of implant abutment, particularly for 28 months of observation. The original clinical case of a custom-made, all-ceramic zirconia Implant Crown device to remove a single tooth was presented in the literature by Kohol and Klaus [151]. Butz et al. [152] compared improvised titanium-reinforced zirconia and natural alumina abutments for their outcomes under fatigue and static loading. The findings revealed that titanium-reinforced zirconia abutments exhibited the same actions as metal abutments. Considering titanium-enhanced zirconia abutments as an esthetic alternative to restaurants of the different implants in the anterior area, the authors recommended using titanium-enhanced zirconia abutments. The efficiency of different implant-zirconia composites under fatigue in an in vitro test was evaluated by Nguyen et al. [153]. They found that the rotational load fatigue of the zirconia abutments was related to the abutment’s diameter. However, various clinical trials have shown the excessive durability of zirconia as a part of the implant and dental prostheses. However, a hindrance to veneering porcelain fracturing is the therapeutic success of a zirconia-based implant. Technical problems are the reason for zirconia fissures [154,155]. In addition, the zirconia durability for a long time is questionable; thus, the ability of titanium to replace zirconia is questionable [156,157].
Esthetic orthodontic brackets centered in zirconia
Besides the previously mentioned dental roles, zirconia was often used to construct esthetic orthodontic braces [158]. Zirconia polycrystalline brackets, the highest strength of all ceramics, were proposed for alumina ceramic brackets. However, for this reason, they are cheaper than alumina ceramic monocrystalline brackets. Shows natural color. Features like large sliding with stainless steel arc-nickel-titanium wires with reduced plaque cohesion, clinical bond strengths, and bond failure sites at the bracket/adhesive joint have been described [159]. However, it can be said that Keith et al. [160] did not find the advantage of zirconia bracket over polycrystalline alumina bracket in terms of friction properties.
Mechanical properties of orthodontic adhesive due to the effects of adding zirconium oxide nanoparticles titanium dioxide
When the orthodontic bracket ligation system is defeated, this will cause the orthodontic brackets to come off and the treatment results to be delayed. About 5-7% of all clinical fractures can occur for various reasons. Several tooth-related or component-related elements can influence the connecting systems and the extent to which orthodontic brackets fail [161]. The shear bond intensity of orthodontic composite resin is greater than that of polyacid resin and resin-adjusted glass ionomer cement [162]. According to other studies, the shear bond strength of orthodontic brackets did not change significantly when examined at various intervals after bonding [163].
In earlier studies, resin monomers, the pretreating of inorganic fillers, and the development of healing technologies have been used to build features of resin-based composites. Heat treatment during and after baking increases both degrees of polymerization and the composite’s strength [164]. Reinforcing fillers for dental composites, fibers, and nanofillers have been proposed, the addition of which increases the composite’s intensity [165]. Much attention has been paid to using fillers (nano-sized) to support prosthetic base resins as advances in nanotechnology. As a result, there has been some thought to making polymer nanocomposites with better mechanical and physical characteristics than nanocomposites containing micro-scale particles. Therefore, composites with significantly enhanced performance can be achieved by using many nanofillers instead of a single additive [166]. Since the mechanical properties of the final polymer nanocomposite are not heavily dependent on the filler's dispersion and adhesion to the matrix, it is essential to treat the fillers' surfaces with a silane coupling agent to establish stability between the two [167]. It is stated that the various properties of nanostructured metal oxides (ZrO2: TiO2) in the mixed form are much higher than their single aggregation due to the size variation between zirconium and titanium. They have amazing physical, mechanical, and photocatalytic properties [168]. Various materials (dodecyl amine, tannic acid derivatives, bipyridine, polyhexane, amphiphilic lipids, silver chlorhexidine gluconate, and fluorides) for long-term sterilization of the area around cohesive restorations that have antibacterial properties have been added over time to dental composite resins. It also combines TiO and fluorides. However, if orthodontics is cohesive, it can improve its antibacterial properties. Therefore, resin-based adhesives' tensile, compressive, and shear bond strengths can be enhanced in vitro using ZrO2-TiO2 nanoparticles [169].
Future trends of zirconia
Biomedical zirconia may be one of the components in orthopedics that sparked debate among scientists, industrialists, and clinicians. The complication of alumina brittleness and resulting capability loss of implants was addressed by the biomedical category zirconia 20 years ago [170]. More than 600,000 femoral zirconia heads have been inserted, mainly in the United States and Europe. On the one hand, the most vital mechanical properties of oxide are shown by biomedical-grade zirconia. On the other hand, if exposed to water, zirconia, due to its ability, will be prone to aging [171]. Some also believe that zirconia itself permits a broad variety of designs. They correctly assert that the degree of defeat before 2001 was very low, and the vulnerability of aging is controlled and minimized by careful monitoring. Others correctly claim that implants with substances not entirely constant in the body are insufficient. Until now, there are already considerably opposite clinical reports: After many in vivo experiments, some findings indicate great behavior of some heads [172]; although some display bad results, the implant is exclusively worn and osteolysis. Given the tightening of alumina and the issue of zirconia aging, a movement is now being created to improve alumina-zirconia composites. This would be the method of toughening zirconia without the key shortcoming correlated with its transfer to steam or body fluid. Several investigations have been performed in the literature concerning zirconia-alumina composites from zirconia-rich and alumina-rich composites [130]. Innovative ceramic firms are designing better products. The composite part is designated to have excellent mechanical and tribology characteristics and is treated with 80% ZrO2-TZP and 20% alumina. The Al2O3-toughened zirconia (ATZ) biomaterial (defined by Metoxit AG) is perfect for sustaining loads four times higher than normal Al2O3. This invention has not been launched yet [173,174].
Conclusion
Because of the transmutation toughening circumstance, zirconia ceramics have been evaluated as the strongest and toughest in comparison with other available dental ceramics. The flexural intensity of zirconia ranges between 800 and 1500 MPa in zirconia partially stabilized structure, as the wholly stabilized structure provides lesser flexural strengths (about 600 MPa). The zirconia fracture toughness ranges between 6.3 and 11.5 MPa m1/2. As for other features such as chemical solubility, zirconia is so permanent, which is significant for withstanding several environmental alterations in the oral environment. Zirconia is characterized by poor thermal conductivity that protects the pulp from temperature alterations in the mouth. Although there are limitations, there is a tremendous need for more comprehensive information regarding aging problems. Regarding dental implants, zirconia is an advanced technology. Also, aging is not a relevant problem for these functions.
Funding
The authors gratefully acknowledge the Research Council of Kermanshah University of Medical Sciences (Grant no. 4010449) for financial support.
Authors' Contributions
All authors contributed to data collection, drafting, and revising of the manuscript.
Conflict of Interest
The authors declare no conflict of interest.
Orcid:
Nafise Nikkerdar: https://orcid.org/0000-0002-2477-1761
Amin Golshah: https://orcid.org/0000-0002-8167-7632
Mohammad Salmani Mobarakeh: https://orcid.org/0000-0002-3272-4041
Nima Fallahnia: https://orcid.org/0000-0002-9571-6137
Bahram Azizi: https://orcid.org/0000-0001-8107-3284
Ehsan Shoohanizad: https://orcid.org/0000-0003-2596-8918
Razieh Souri: https://orcid.org/0000-0002-3960-2522
Mohsen Safaei*: https://orcid.org/0000-0003-3885-6640
--------------------------------------------------------------------------
How to cite this article: Nafise Nikkerdar, Amin Golshah, Mohammad Salmani Mobarakeh, Nima Fallahnia, Bahram Azizi, Ehsan, Shoohanizad, Razieh Souri, Mohsen Safaei, Recent progress in application of zirconium oxide in dentistry. Journal of Medicinal and Pharmaceutical Chemistry Research, 2024, 6(8), 1042-1071. Link: https://jmpcr.samipubco.com/article_192158.html
--------------------------------------------------------------------------
Copyright © 2024 by SPC (Sami Publishing Company) + is an open access article distributed under the Creative Commons Attribution License(CC BY) license (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

.png)
.png)
.png)
.png)
.png)
