Document Type : Case Report
Authors
Department of Pulmonology and Respiratory Medicine, Faculty of Medicine, Universitas Airlangga/Dr. Soetomo General Hospital, Surabaya, Indonesia
Abstract
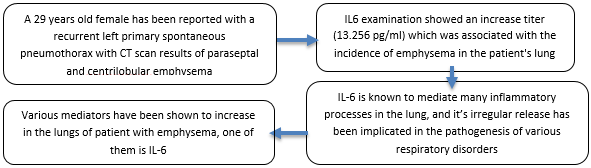
Primary spontaneous pneumothorax (PSP) is characterized by the occurrence of pneumothorax in the absence of any underlying lung disease. The pulmonary abnormalities found in patients with PSP have been identified as emphysema-like such as blebs and bullae. Even though until now, the mechanism of the immune response related to the formation of emphysema is still unclear, several studies have investigated the effect of cytokines on the incidence of emphysema, one of them is the IL-6 cytokine group. A 29 years old female has been reported with a recurrent left primary spontaneous pneumothorax with CT scan results of paraseptal and centrilobular emphysema of the superior and middle lobes of the right lung. IL6 examination showed an increase titer (13.256 pg/ mL) which was associated with the emphysema incidence in the patient's lung. IL-6 is a substance activated by T and B lymphocytes, monocytes, fibroblasts, and activated macrophages. This cytokine is recognized for its role in mediating numerous inflammatory processes within the lung. The erratic secretion of IL-6 has been associated with the development of various respiratory conditions, encompassing asthma, COPD, and idiopathic pulmonary fibrosis (IPF). Various mediators have been shown to increase in the lungs of patient with emphysema; one of them is IL-6. Further research is needed to get the benefits from the direct anti-IL-6 therapy in emphysema.
Graphical Abstract
Keywords
Main Subjects
Introduction
Spontaneous pneumothorax is characterized by the presence of air in the pleural cavity, not attributable to trauma or iatrogenic causes [1]. Spontaneous pneumothorax is categorized into primary spontaneous pneumothorax (PSP) and secondary spontaneous pneumothorax (PSS) [2], depending on whether there is clinically apparent underlying lung disease [3]. Other literature also explains that PSP, occurring in individuals without pre-existing lung disease, is thought to stem from the rupture of subpleural emphysema blebs, frequently situated at the apex of the lung [4]. Although the mechanism of the immune response related to the formation of emphysema is still unclear, several studies have examined the influence of cytokines on the incidence of emphysema, one of which is the IL-6 cytokine group. Increased IL-6 expression and IL-6 gene polymorphisms are associated with a rapid decline in lung function and increased disease risk in smokers [5].
Case Illustrations
A 29 year old woman with a left primary spontaneous pneumothorax who had a chest tube inserted, two weeks after the chest tube was installed, her lungs were declared fully expanded and the chest tube was removed. However, 1 week later the patient complained of shortness of breath again, and found a left recurrent spontaneous pneumothorax, chest tube insertion and pleurodesis were performed twice, but it did not improve, timeline of chest X ray can be seen in Figure 1. Currently the patient is pregnant with a gestational age of 20 weeks. The patient works as a state civil servant in his city. A history of smoking was denied, but the patient's brother and husband smoked. Her mother and brother had history of pneumothorax and had also a chest tube inserted.
The general condition appears to be moderately ill, with overweight nutritional status, weight 73 kg and height 158 cm. Glasgow coma scale 4-5-6, blood pressure 120/70 mmHg, pulse 110 times/minute, breathing 24 times/minute, axillary temperature 36 °C, oxygen saturation 93-94% with room oxygen. On physical examination of the lungs, left asymmetric movement was left behind, palpable fremitus decreased in the left hemithorax, sonor in the right hemithorax, hypersonor in the left hemithorax, and vesicular breath sounds decreased in the left hemithorax, rhonchi and wheezing were not found in both lung fields. Other physical examinations revealed no abnormalities.
Patient underwent a chest CT and emphysema like-changes was found with left fluidopneumothorax which can be seen in Figure 2, and for laboratory testr showed an increase in IL-6 as a pro-inflammatory cytokine (Table 1).
Discussion
PSP is a thoracic disease that most often attacks adolescents and young adults [6]. In this case, the patient was suspected of PSP due to his young age, 29 years, apart from that the patient did not have any other diseases that could cause pneumothorax. Some evidence supports a genetic contribution to pneumothorax. Mainly familial clusters, observed in 10-12% of cases, and gene mutation findings in familial and sporadic cases. Moreover, several Mendelian disorders, such as Birt-Hogg-Dubé and Marfan syndromes, manifest pneumothorax as a clinical feature [1]. The clinical course of PSP varies with recurrence rates ranging from 17% to 54% [2].
Pathogenesis PSP
Although PSP is usually defined as a pneumothorax without any lung abnormalities, it is increasingly evident that these patients do not have “normal lungs” [7]. In contrast to the defined criteria, CT scans often reveal pleural pathology, such as blebs/bullae or emphysema-like changes, in 63-80% of PSP patients [8]. Emphysema is generally classified based on the part of the acinus affected, namely panacinar (panlobular) emphysema, centriacinar (centrilobar) emphysema, distal acinar (paraseptal) emphysema, and irregular emphysema associated with fibrosis [9,10]. Rupture of subpleural blebs or small bullae stands out as the leading cause of PSP. However, it is still not certain how bullae, which are key to the pathogenesis of PSP, form in the lungs of young patients and healthy lungs [11].
Various factors, including hereditary predisposition, abnormal connective tissue, low body mass index, caloric restriction, apical ischemia of the apex, anatomical abnormalities in the bronchial tree, ectomorphic physiognomy with increased negative intrapleural pressure, and distal airway inflammation, can be associated with the development of blebs, bullae, and areas of pleural porosity [12,13]. The lungs can suffer damage and inflammation from inhaling cigarette smoke and other toxic particles, resulting the emergence of airway diseases such as bronchiolitis and chronic bronchitis and in the destruction of lung parenchyma (emphysema).
The role of IL-6 in inflammation of the lung
Activated by T and B lymphocytes, monocytes, fibroblasts, and macrophages, IL-6 is a substance produced in response to various immune system elements [14]. IL-6, Known for its role in mediating numerous inflammatory processes in the lungs, has been associated with the development of various respiratory disorders when released in a dysregulated manner [15,16]. A potential link between IL-6 and the onset of pulmonary fibrosis is indicated by various genetic studies in both animals and humans. Furthermore, concentrations of IL-6 in BAL fluid were found to be 100 times higher in patients with ARDS compared to normal subjects, persisting above normal levels for as long as 21 days [17]. Another study reported that IL-6 levels in sputum were increased in COPD patients, as well as in the BAL fluid of pulmonary emphysema patients [16].
IL-6, functioning as an inflammatory and pro-fibrotic cytokine, plays a role in the pathogenesis of COPD, idiopathic pulmonary fibrosis (IPF), and lung diseases like asthma [18]. The excessive endogenous production of IL-6 in the lungs, as demonstrated by Ruwanpura et al., is implicated in the development of emphysema. In the lungs, the elevated upregulation of IL-6 was found to lead to increased pulmonary apoptosis, a process likely involved in the pathogenesis of IL-6-induced emphysema, as revealed by the study [19].
Furthermore, the link between the increased expression of IL-6 in mouse models of emphysema and the pathogenesis of this disease has been established [5]. Although the animal model used in the study is less informative for genetically explaining the cellular and molecular events of IL-6 in pathogenesis, the induced over-expression of IL-6 and IL-11 in the lungs of transgenic mice leads to dilation of the alveoli, accompanied by infiltration of mononuclear inflammatory cells [19, 20]. These cytokines elicit a cellular response through the signal transduction receptor gp130, facilitating Janus kinase (Jak)–Signal transducer activation [5,19]. While other IL-6 cytokine groups transmit signals via the heterodimerization of gp130 with other chain structures, IL-6 and IL-11, in particular, initiate intracellular signaling through the homodimerization of gp130. Enlargement of alveolar cavities is observed in transgenic mice with artificial, lung-specific overexpression of IL-6 or IL-11, supporting the implication of a hyperactive gp130 signaling pathway in the development of emphysema [19]. With IL-6 levels in this patient showing an increase of 13,256 pg/mL, it can be inferred that the elevated IL-6 levels were associated with the occurrence of emphysema detected in chest CT scans.
Conclusion
PSP is a thoracic disease that most often attacks teenagers and young adults. In most patients with PSP, chest CT scan findings show emphysema-like changes. One case of a 29-year-old woman with recurrent spontaneous penumothorax has been reported. Patients with a history of previous PSP and who have had pleurodesis but have not improved. The occurrence of recurrent pneumothorax accompanied by pulmonary emphysema in this patient was associated with a high increase in IL6 levels in the blood, which can be caused by repeated exposure to cigarette smoke both at home and in the work environment.
Acknowledgements
This case report was made possible thanks to assistance from various parties, for this reason the researcher would like to thank Elisabeth Tri Wahyu W and Devi Ambarwati who have contributed to the examination of IL6 and MMP9 in the case report.
Funding
No funding was received for this study.
Authors’ contributions
All authors contributed toward data analysis, drafting, and revising the paper, gave final approval of the version to be published, and agreed to be accountable for all aspects of the work.
Conflict of Interest
The authors declare no conflict of interest.
Orcid:
Phutri Pratiwy: https://orcid.org/0009-0009-7826-3066
Daniel Maranatha*: https://orcid.org/0000-0002-4908-9005
--------------------------------------------------------------------------
How to cite this article: Phutri Pratiwy, Daniel Maranatha, The role of IL-6 in pulmonary emphysema with recurrent primary spontaneous pneumothorax in a young woman: a case report. Journal of Medicinal and Pharmaceutical Chemistry Research, 2024, 6(8), 1142-1148. Link: https://jmpcr.samipubco.com/article_193194.html
--------------------------------------------------------------------------
Copyright © 2024 by SPC (Sami Publishing Company) + is an open access article distributed under the Creative Commons Attribution License(CC BY) license (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

.png)
.png)
.png)
