Document Type : Original Research Article
Authors
- Quang Hien Tran 1, 2
- Van Binh Phan 3
- Hoang Phuong Ho 3
- Anh Hien Ho-Thi 3
- Tin Trung Nguyen 4
- Quang Huy Huynh 5
1 An Giang Department of Health, An Giang, Vietnam
2 Department of Obstetrics and Gynecology, An Giang Women and Children’s Hospital, An Giang, Vietnam
3 Department of Radiology, Tam Anh General Hospital, Ho Chi Minh, Vietnam
4 Department of Radiology, Ho Chi Minh City Oncology Hospital, Ho Chi Minh City, Vietnam
5 Department of Radiology, Pham Ngoc Thach University of Medicine, Ho Chi Minh city, Vietnam
Abstract
To assess the depth of myometrial invasion in patients with endometrial cancer by contrasting the diagnostic efficacy of dynamic contrast enhanced (DCE) magnetic resonance imaging with diffusion-weighted (DWI) MRI. 64 Patients with endometrial cancer who had preoperative 3.0 Tesla MRI in sagittal planes, including T2-weighted (T2W), DWI (b=0 and 1000 s/mm2), and dynamic contrast enhanced (DCE) MRI, were retrospectively evaluated. Results of surgical pathology were linked with the depth of myometrial invasion on MRI. A radiologist determined the definitive histologic grade (G1-G2; G3) and compared it with the mean ADC of the tumor and peritumoral zone. The evaluation of myometrial invasion was enhanced when using DWI or DCE imaging values were combined with T2W imaging. Sensitivity, specificity, accuracy, positive predictive value, negative predictive value, and area under the receiver operating characteristic (ROC) curve were measured for determining the depth of myometrial invasion: T2W-DWI / T2W-DCE imaging; 96.15%/92.31%, 85.19%/85.19%, 90.57%/86.68%, 86.21%/85.71%, 95.81%/92%, and 0.91/0.89. The average ADC offered accurate and valuable information for estimating the tumors' histopathological grade. The area under the ROC curve, sensitivity, specificity, accuracy, negative predictive value, and positive predictive value in our series were 64%, 87.18%, 76.19%, 79.07%, 78.13%, and 0.763, respectively, for a cutoff value of 0.638 x 10-3 mm2/s. T2W-DWI is a potential replacement for DCE in the assessment of the depth of myometrial invasion of endometrial cancer, especially useful for patients with contraindications to contrast agents. The combination of T2W and DWI has high diagnostic high accuracy in the assessment of the depth of myometrial invasion.
Graphical Abstract
Keywords
Main Subjects
Introduction
The most common genital cancer in women in developed countries is endometrial carcinoma, which is the second most common globally after cervical carcinoma [1]. The histological type, tumor grade, and International Federation of Gynecology and Obstetrics (FIGO) stage at diagnosis- specifically, the depth of myometrial invasion- all affect the prognosis of endometrial cancer [2]. Planning treatment and referring patients to gynecologic oncologists can benefit from accurate preoperative assessment of the tumor's extent. Individuals with grade 3 endometrioid tumors, deep myometrial invasion, cervical stromal involvement, and non-endometrioid histologic subtypes of endometrial cancer are among those who have an increased risk of lymph node metastases. These patients ought to have their lymph nodes thoroughly dissected [3]. The patient's surgical approach is influenced by preoperative risk classification; in situations of grade 1 tumors and penetration of less than 50% of the muscle layer thickness, surgeons do not need to do lymph node dissection.
At the moment, deep MRI, including T2-weighted (T2W) and dynamic contrast-enhanced (DCE) imaging, can detect myometrial invasion in women with endometrial cancer preoperatively [4]. According to certain research, DWI is superior to DCE-MRI for evaluating endometrial cancer's myometrial invasion [5]. Other research, however, showed no distinction between diffusion-weighted imaging (DWI) and dynamic contrast enhanced (DCE) MRI. It appears that the DWI inclusion enhances the preoperative diagnostic precision of MRI in determining the extent of myometrial invasion in cases of endometrial cancer. For patients for whom contrast agents are contraindicated, this could be useful. This study aimed to compare the diagnostic efficacy of DCE MRI and DW MRI in assessing the overall stage and depth of myometrial invasion in endometrial cancer patients.
Experimental
Patient selection
This was a retrospective study conducted at a single center on 64 women, ages ranging from 37 to 76 years. Every woman had received pelvic MRI, including T2W, DCE, and DWI, and had surgically proven primary endometrial cancer. Patients who have received surgery or preoperative MRI at another facility were not accepted.
Surgeons with over ten years of experience in treating gynecological cancers, conducted surgical procedures in a specialist oncology center. Following the preoperative MRI, all procedures took place between one and thirty-four days. The diagnostic benchmark for comparison was the outcome of the histological examination, which was carried out by a pathologist with over 20 years of experience and specialization in gynecology/oncology.
MRI protocol
A 3.0 Tesla MRI scanner (GE Signa HDxT 3T) was used to do the MRI. Before the evaluation, patients were instructed to fast for four hours. An intramuscular injection of N-butylscopolamine bromide (20 mg) was given before the MRI to decrease peristaltic artifacts and bowel motility. Patients were made to lie supine during the examination. Table 1 indicates the MRI methodology used in the study.
Image analysis
Two radiologists with three and twenty years of experience, respectively, specializing in oncology radiology, assessed the MRI scans and determined the tumor stage and depth of myometrial invasion using the FIGO staging system, taking into account three sets of sequences: T2WI, DCE+T2WI, and DWI+T2WI. Both radiologists were blinded to the histopathological reports. The T2WI sequence was analyzed first in each patient, and the depth of myometrial invasion was assessed. Next, the DCE+T2WI and DWI+T2WI sequences were analyzed. T1-weighted imaging was examined separately in each instance to rule out any possible dangers, like bleeding. If the tumor had invaded less than 50% of the myometrial thickness, the invasion was categorized as "superficial," and if it had penetrated more than 50%, it was classed as "deep".
Histological findings
Complete hysterectomy, bilateral salpingo-oophorectomy, pelvic lymphadenectomy, and peritoneal lavage were performed on each patient. The tumor's histological type, grade, and level of myometrial invasion (deep or superficial) were all evaluated histopathologically.
We took into consideration two factors to ascertain the depth of myometrial invasion. Taking into account the parts that were not penetrated, we first computed the total thickness of the myometrium. Next, we ascertained if the tumor cells had infiltrated the vascular plexus that divides the two strata.
Statistical analysis
Version 20.0 of the SPSS software was used for all statistical analyses. The data are shown as proportions, absolute and relative values, or 95% confidence intervals (95% CI). For T2WI, DCE+T2WI, and DWI+T2WI, the diagnostic accuracy, sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) were computed.
Results
Table 2 provides an overview of the patient's demographics and postoperative histology results. 31 (57%) and 33 (43%) of the 64 patients, respectively, showed superficial and profound myometrial invasions.
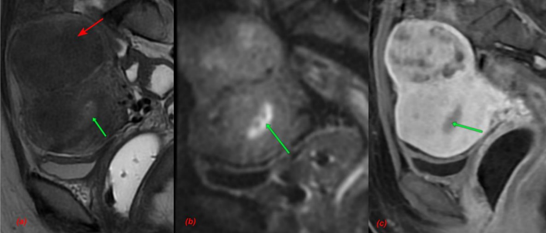
Of the 64 patients; the depth of myometrial invasion was appropriately assessed by T2WI in 56 (82.05%), DCE + T2WI in 60 (92.19%), and DWI + T2WI in 59 (93.75%) (Table 2). 22 patients (50%) had correct staging determined by T2WI, 34 (77%), by DCE-MRI+T2WI, and 41 (93%) by DWI+T2WI. When evaluating the depth of myometrial invasion, DWI+T2WI outperformed T2WI and DCE-MRI+T2WI in terms of diagnostic accuracy, sensitivity, specificity, PPV, and NPV (Table 3). The MRI of a patient with endometrial cancer and concurrent leiomyoma is displayed in Figure 1. Figure 2 depicts concomitant adenomyosis and endometrial carcinoma. Table 4 shows no statistically significant correlation between ADC levels and myometrial invasion. Table 5 shows a significant difference (p=0.003) in the ADC mean value of the tumor region between the high-grade (G3) and low-grade (G1-G2) groups. The tumor region's ADC mean value may distinguish between the G1-G2 and G3 groups, as shown by the area under the receiver operating characteristic curve (AUC) of 0.763. A cutoff of 0.638 x 10-3 mm2/s was employed at Youden index J = max (Se + Sp - 1) to distinguish between the G1-G2 and G3 groups; the corresponding sensitivity, specificity, PPV, NPV, and accuracy were 64%, 87.18%, 76.19%, 79.07%, and 78.13% (Figure 3).
Discussion
Value of MRI sequences in assessing myometrial invasion
For women with endometrial cancer, MRI with T2W and DCE sequences is currently thought to be the gold standard for assessing myometrial invasion before surgery [6]. Nevertheless, the T1W sequence necessitates the intravascular injection of a contrast agent, which may not be appropriate for certain women experiencing severe renal failure, medication allergies, or pregnancy. Disruption of the transition zone indicates the presence of myometrial invasion. The endometrium has a high signal intensity on T2W, while the transition zone has a low signal intensity. The accuracy of this approach can be decreased by several concomitant circumstances, including an indistinct transition zone (common in post-menopausal women), poor contrast between the tumor and myometrium, and constriction of the uterus by fibroids, adenomyosis, or polypoid tumors [7]. Small tumors confined to the endometrial cavity can be identified by DCE because the endometrial tumor absorbs the contrast agent in about 30 seconds (during the arterial phase, earlier than the normal endometrium). In addition, compared to the uterine muscle, the tumor absorbs less of the contrast agent. Around 120-180 seconds after injection, during the equilibrium period, there is the greatest contrast between the high signal intensity of the healthy endometrium and the reduced signal intensity of the tumor [8]. Furthermore, the DCE accuracy may be lowered by uterine fibroids, adenomyosis, tumors growing into the uterine horn, and inflammation-related contrast agent capture [4]. In our investigation, the T2W pulse sequence's accuracy, sensitivity, specificity, PPV, and NPV in determining deep muscle invasion were 83.02%, 96.15%, 70.37%, 75.76%, and 95%, in that order. These rates were 95.83%, 96.15%, 85.19%, 90.57%, and 86.21% when paired with DWI, in that order. These rates increased to 86.68%, 92.31%, 85.19%, 86.21%, and 95.83% when DCE was added. Our research validates the usefulness of CHT in evaluating muscle invasion in endometrial cancer. Compared to the T2W pulse sequence, T2W-DWI and T2W-DCE have superior diagnostic capabilities. There was no statistically significant difference between T2W-DWI and T2W-DCE when it came to evaluating deep muscle invasion of UTBM NMTC. However, T2W-DWI had a little higher accuracy than T2W-DCE. This outcome aligns with the findings of previous research conducted across the globe. As per the meta-analysis conducted by Andreano et al. [9], the DWI pulse sequence has a high value in diagnosing the degree of muscle invasion and there is no significant difference between the DWI and DCE pulse sequences [10]. In the meta-analysis by Deng et al. [11], T2W-DWI's (0.858) sensitivity was comparable to T2W-DCE's (0.863). However, T2W-DWI's (0.947) specificity was greater than T2W-DCE's (0.865) (p=0.028). Consequently, in the preoperative assessment of patients with renal impairment or contraindications to contrast agents, T2W-DWI can take the role of DCE.
Significance of ADC in the tumor region and the surrounding region in assessing myometrial invasion and predicting the histologic grade of endometrial cancer
According to our analysis, the tumor region's mean ADC value was 0.7x10-3 mm2/s. Groups with varying histological grades showed significant differences in the ADC mean values of different uterine areas (p=0.002). Additionally, the results of our study demonstrated that the G1-G2 group and the G3 group could be distinguished from one another based on the ADCmean values of the uterine areas, with the G1-G2 group having higher ADC mean values. To distinguish between these two groups, we set a threshold ADCmean value of 0.59 x10-3 mm2/s. The corresponding values for sensitivity, specificity, PPV, NPV, accuracy, and AUC were 57.89%, 91.17%, 78.57%, 79.48%, 79.25%, and 0.754. Studies that used group divisions comparable to ours also discovered statistically significant variations in ADC values. Reyes-Pérez et al. (2020) [12] detected lower ADCmin and ADCmean values in the G2-G3 group than in the G1 group. To distinguish between the G1-G2 and G3 groups, the ADC min and ADC mean values were, respectively, 0.69 x10-3 mm2/s (sensitivity, 96%; specificity, 68%) and 0.82 x10-3 mm2/s (sensitivity, 96%; specificity, 58%). ADC mean's AUC was 0.8 while ADC min's was 0.82.
Other studies, such as that by Seo [13], used different group divisions and also found differences in the ADCmean values of the uterine regions between the G1 and G2-G3 groups.
However, because different histological grades have overlapping values, using ADC values to predict the histological grade of uterine cancers before surgery has limits. The uterus's histological grade is not solely determined by cell density; other crucial traits, such as nuclear deformation, are also present but cannot be seen with traditional histological examination [14]. Tumor growth, blood perfusion, tissue buffering, and other factors can all have an impact on ADC readings in addition to the uterine cell density [15]. Furthermore, some research has demonstrated that variations in cell membranes and stages of cell division can affect how water molecules flow [15,16]. The final ADC value is determined by combining all these factors, which may explain the overlapping ADC values.
Conclusion
Combining T2W with the DWI and DCE pulse sequences enhances the diagnostic value of assessing deep tumor infiltration. The sagittal DWI pulse sequence can replace DCE in evaluating infiltration in patients with contraindications for contrast agents without compromising diagnostic value. Using a cutoff point of 0.59 x 10-3 mm2/s for the ADCmean value of the uterine regions, the sensitivity, specificity, accuracy, PPV, and NPV for differentiating between the G1-G2 and G3 uterine groups were 57.89%, 91.17%, 79.25%, 78.57%, and 79.48%, respectively.
Ethical approval and informed consent
All methods were conducted according to the ethical standards of the Declaration of Helsinki. Ethical clearance was obtained from an institutional Research Ethics Review Board (IRB) of Pham Ngoc Thach University of Medicine (reference number 770/PNTUM-EC) and the IRB of HCM Oncology Hospital (reference number 208/HCMOH-EC). Written informed consent was obtained from the participants. There are no study participants under the age of 18 years.
Acknowledgements
We would like to thank everyone who supported and helped us to complete this study.
Funding
This study did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors' Contributions
All authors contributed equally to this work, they contributed to data analysis, drafting, and revising of the manusrcipt and agreed to be responsible for all the aspects of this work.
Conflict of Interest
No potential conflict of interest was reported by the authors.
Orcid:
Quang Hien Tran: https://orcid.org/0000-0003-4166-4076
Van Binh Phan: https://orcid.org/0000-0001-8623-0314
Hoang Phuong Ho: https://orcid.org/0009-0003-6124-4834
Anh Hien Ho-Thi: https://orcid.org/0000-0003-3067-2468
Tin Trung Nguyen: https://orcid.org/0009-0008-5041-3627
Quang Huy Huynh*: https://orcid.org/0000-0001-5177-1460
----------------------------------------------------------------------------
How to cite this article: Quang Hien Tran,Van Binh Phan, Hoang Phuong Ho, Anh Hien Ho-Thi, Tin Trung Nguyen, Quang Huy Huynh, The role of 3.0 tesla diffusion-weighted magnetic resonance imaging in evaluating the depth of myometrial invasion and predicting the histological grade of endometrial cancer. Journal of Medicinal and Pharmaceutical Chemistry Research, 2024, 5(9), 1469-1477. Link: https://jmpcr.samipubco.com/article_194932.html
----------------------------------------------------------------------------
Copyright © 2024 by SPC (Sami Publishing Company) + is an open access article distributed under the Creative Commons Attribution License(CC BY) license (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

.png)
.png)
.png)
.png)
.png)
.png)
